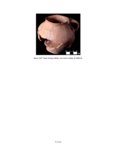
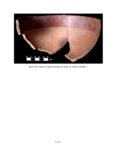
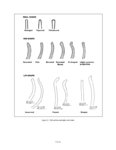
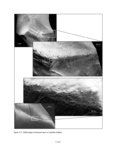
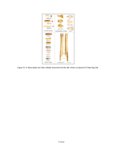
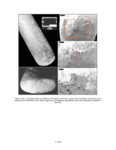
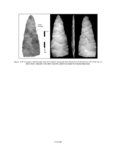
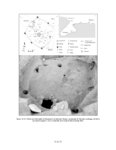
| OCR Text |
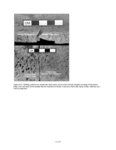
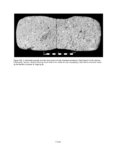
Show 2 and juniper boughs used for roof thatch), firewood gathering, ceremonial or ritual activities (e.g. scattering corn pollen or pollen face paints), and food processing. As with macrobotanical analyses, archaeological pollen records contain a history of accidents and events. The recognition of cultural pollen signals is usually, depending on the integrity and type of sample context, compromised by the influence of the natural pollen rain. The best criteria for interpreting past cultural activities is when pollen is represented over what would be expected from natural background pollen rain or when there are repeated associations of pollen types or suites of types by context (Bohrer 1981). METHODS Three types of samples were collected and processed for the N16 project: small pieces of moss called polsters (n = 4), which are often used to establish modern pollen analogs in paleoenvironmental reconstructions (Moore et al. 1991:36); bulk sediment samples both from the modern ground surface (n = 5) and from buried contexts (n = 220); and pollen washes of seeds and artifacts. The extraction and analysis methods used on the pollen washes are documented in Chapter 12. All of the bulk sediment samples were processed by the following procedure. Subsamples, typically 20 cc, but occasionally as small as 10 cc when little sediment was available, were taken from the sample bags and spiked with a known concentration of tracer tablets (Lycopodium spores). Lycopodium tracers enables calculation of pollen concentrations, which are estimates of the absolute abundance of pollen in each sample, expressed as numbers of pollen grains per cubic centimeter of sample sediment, abbreviated as gr/cc. Hydrochloric acid (10% solution) was added to the sediments to dissolve caliche and carbonates, followed by sieving through a 0.18 mm mesh stainless steel screen to separate coarse sediment and other macro materials. Samples were then mixed with a zinc bromide heavy liquid (specific gravity 2.0) and the lighter particles, including pollen, were floated from the heavy inorganic fraction. Acetolysis, a chemical technique that digests organic materials (except pollen), was used on the samples next, followed by a 10minute hot hydrofluoric acid treatment (approximately 49% solution), which dissolves silicate minerals. The final extracted residues were rinsed with alcohol, mixed with glycerol, and stored in glass vials. The N16 pollen samples were all processed before 1996, and the extraction procedure described above was the best known process at the time. Excluding pollen washes and modern control samples, which are special contexts, 49 of the 220 bulk sediment samples did not produce adequate pollen for a significant count. The extraction procedure at the Laboratory of Paleoecology, Northern Arizona University was significantly changed in 1997, based on experimental data (Smith 1998), and additional refinements were added in 2000. It is likely that fewer sterile samples would have resulted with use of the new extraction procedures. Moss polster samples were processed by the same method as the bulk sediment samples, except no heavy liquid density separation was used, and before the hydrochloric acid step, the polsters were washed with a 10 percent solution of potassium hydroxide, which acts as a dispersant. Two levels of microscopy were used to identify the pollen assemblages from the modern controls and archaeological samples: 400x magnification counts of all pollen types to 200 or more grains, if possible, and 100x magnification scans for larger pollen types. If preservation is moderate, pollen greater than 30 µm in size is easily identified at 100x magnification; this includes corn, squash, cacti, and some herb types. Aggregates (clumps of the same pollen type) were counted as one grain per occurrence, and the taxon and number of grains in the aggregate were recorded separately. Numerous large aggregates in protected archaeological contexts can reflect plant processing. Pollen identifications were made to the lowest taxonomic level possible, based on published keys (Kapp et al. 2000; Moore et al. 1991) and the Laboratory of Paleoecology pollen reference collection at Northern Arizona University (http://www4.nau.edu/becbase/). Sunflower family pollen (Hi-Spine Asteraceae) was differentiated from the ragweed type (Low-Spine Asteraceae) based on spine height greater than 2 µm, as proposed by Hevly et al. (1965). Pinyon pine was separated from other pines by the length of grains, based on keys developed by Jacobs (1985). Discussion of plants and pollen types entails the language of plant names, which can be confusing, and the topic is further complicated by the fact that pollen names are not directly equivalent to plant names. Pollen taxa typically subsume several plant species and genera, as it is rare that an individual plant species is represented by a pollen type. Another aspect of botanical confusion is the fact that the scientific nomenclature is perpetually under construction as species are reevaluated, and the pace of taxonomic change has increased with the advent of DNA studies. In this chapter, the following conventions are used to provide consistency. Pollen types are discussed by the common names; these are listed in Table 11.2 with both the common and scientific names for reference. If a plant name is V.1.2 |