| OCR Text |
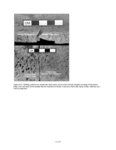
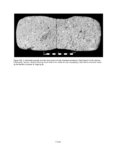
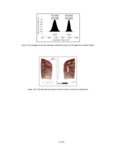
Show elements in the upper range of trace concentrations, but more typically it is used to characterize major and minor elements in a sample. Sensitivity is affected by the detection limits of the specific machine, which are a function of count time. The microprobe used for this analysis, a Cameca Camebax MBX, can routinely analyze elements with an atomic number greater than 9 (fluorine), and can also detect carbon, nitrogen, and oxygen with minor adjustments to the crystals. Precision refers to the replicability of an analysis; that is, will the machine consistently produce the same results from analysis of the same sample? Precision of the microprobe is generally quite good, but can be limited by several factors. Poor sample preparation, including inadequately polished thin sections, affects precision through absorption and scattering of X-rays. Poor focus or insufficiently long counting times for elements that are present in low concentrations can also affect precision. Electronic "drift" of the machine, which occurs as the filament generating the electron beam current burns down and deforms, is another concern. To increase precision, the operator must ensure that analytical technique is rigorous and consistent. To enhance the precision of this study, a standard material of known composition was analyzed before and after each sherd or ash sample, and if a discrepancy of more than 5 percent existed, the machine was restandardized. Accuracy refers to how representative the analysis is of the actual elemental concentration in the sample. Accuracy is controlled by calibration of the microprobe using standard material samples with known compositions. Microprobe analysis can consistently detect element concentrations within 1 or 2 relative percent of the actual concentrations. Errors in accuracy are most often due to improper focus on the specimen or inadequately polished slides, which cause the X-rays to be absorbed or scattered. Accuracy may also be affected by the use of incorrect matrix correction (due to inappropriate standard materials) or the use of standards with poorly known compositions. In this study accuracy was assured by vigilant machine operation using standardization materials that are appropriate for characterization of rhyolitic volcanic ash. The microprobe offers a relatively inexpensive method to obtain compositional analyses of ceramics, stone, or metal artifacts, and it provides results more quickly than instrumental neutron activation or other techniques that require extensive sample preparation and analysis time. The main advantage of the microprobe over techniques such as X-ray fluorescence, to which it is quite similar, is that the electron beam can be focused to analyze only a few cubic micrometers (µm) of material. This permits analysis of minute portions of an artifact, such as ceramic temper, which would be difficult to separate from the sample matrix by mechanical methods. The electron microprobe is also useful for archaeological studies because it is nondestructive. Although a sherd or small portion of an artifact must be cut and polished to produce a thin section, the resulting sample can be used for repeated analyses and can be curated for future studies. Additionally, the small size of the thin sections used by the microprobe (1-inch diameter) makes it possible to use pottery sherds or small pieces removed from vessels as samples. ANALYTICAL METHODS The initial goal of the microprobe research was a quantitative compositional analysis of volcanic ash temper in Tusayan White Ware sherds from the Kayenta region and in samples of volcanic ash from Blue Canyon. Elements analyzed with the microprobe were Na (sodium), Mg (magnesium), Al (aluminum), Si (silica), K (potassium), Ca (calcium), Ti (titanium), Mn (manganese), and Fe (iron); all elements are reported as oxide weight percents (wt %) of Na2O, MgO, Al2O3, SiO2, K2O, CaO, TiO2, MnO, and FeO. These elements, as oxides, represent the major constituents of rhyolitic volcanic tephra (Deer et al. 1971; Izett et al. 1988), and are appropriate for characterizing and comparing the composition of volcanic ash deposits. Microprobe Operating Conditions and Procedures Microprobe analysis took place at the Bilby Research Center, Northern Arizona University, in Flagstaff. The Cameca Camebax MBX electron microprobe is equipped with three wavelength-dispersive spectrometers and a Quantum thin-window energy-dispersive spectrometer. Standard 1-inch round thin sections were made from each ceramic sample after photographing the sherds to record their painted designs. After polishing, the samples were coated with a thin (250 Å) layer of carbon, which prevents the sample from becoming electrically charged by conducting electrons to the edge of the section. Operating conditions of the microprobe can be set according to the operator's preferences and the materials being analyzed. In this study, a vacuum of 5 x 10-5 Torr (1 Torr = 1/760 atmosphere) was maintained in the electron beam column and sample chamber. The accelerating voltage was 15 keV (kiloelectron volts) and the specimen current was 10.0 nA (nanoamps). Electron beam diameter was generally 3 µm, small enough to analyze individual fragments of volcanic ash temper but exclude the surrounding V.4.3 |