| OCR Text |
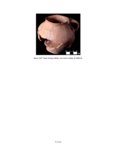
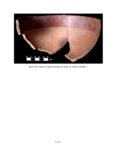
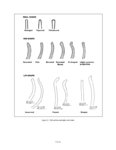
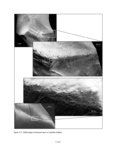
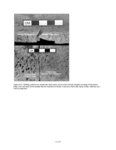
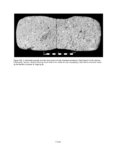
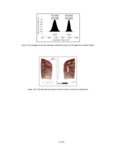
Show sherds, (2) identify production areas for these vessels, (3) conduct field reconnaissance to locate the source(s) of the ash used for ceramic production, and (4) model the exchange network for ash-tempered ceramics in the northern Kayenta region. The microprobe study included Tusayan White Ware ceramics from 11 sites along the road corridor, representing 14 distinct temporal or spatial components. Samples were selected to include the greatest variety of ceramic types and vessel forms from multiple different features at each site. This chapter incorporates results from microprobe analyses of sherds from Kayenta Anasazi sites along Navajo Route 16 (N16) and Navajo Route 21 (N21), and in the Klethla Valley. By including a large number and geographic range of samples, we are better able to discuss models of ceramic production and exchange. Also included are analysis data for samples of volcanic ash collected from Blue Canyon and from two Kayenta sites. INTRODUCTION TO ELECTRON MICROPROBE ANALYSIS The electron microprobe was developed in the late 1940s, and was quickly recognized as a useful tool for chemical characterization of a variety of materials. It is particularly useful for the identification of mineral inclusions in parent material, due to the ability to focus the analysis on a small area. By the early 1960s the microprobe had been adopted by European scientists as a technique to address archaeological problems ranging from identifying the provenience of stone and metal artifacts to characterizing the technologies involved in producing these items. The microprobe was less widely used in the United States until the 1970s, but it has gained popularity in the past several decades. Although the microprobe uses a physical technique (the interaction of electrons), it is considered a type of chemical analysis because it produces a quantitative chemical composition of the sample. A sample of the material to be analyzed is cut to size (for samples containing inclusions of interest) or powdered and mixed with epoxy (for homogeneous samples), and then ground into a polished thin section. The thin section is coated with a thin layer of carbon to prevent the sample from becoming electrically charged. The microprobe produces a beam of electrons that passes through a series of magnetic lenses and strikes the area of interest on the target specimen, interacting with the atoms in the specimen. Inner-shell electrons in the atoms are knocked out of their orbits by the impact of the electrons, and as the resulting ions return to a normal stable energy state, X-rays characteristic of the elements are produced. The X-rays emitted by this process may be detected by either wavelength or energy spectrometers. Wavelength dispersive spectrometry (WDS) counts the emitted X-rays of specific elements and provides a quantitative analysis of those elements in the sample. Energy dispersive spectrometry (EDS) detects the entire range of X-rays emitted by the sample and provides a semi-quantitative analysis; the relative amount of each element, rather than the absolute amount, is displayed. EDS can be completed more rapidly, but is mainly useful for exploratory research because the absolute composition of the sample remains unknown. Birks (1963) and Fitzgerald (1973) provide more detailed descriptions of the mechanical workings of the electron microprobe for the interested reader. Ceramic studies initially focused on the characterization of slips and glazes (Nelen and Rye 1976; Tite et al. 1982), takaing advantage of the microprobe's ability to examine a specific area and depth on the sample, eliminating the problems involved in separating the surface coating from the body of the pottery. The microprobe has increasingly been used to characterize ceramic paste and temper, often in an effort to determine manufacture location. Although some researchers (Abbott 2000) have successfully studied paste composition, Vint (1992) showed that this application can produce inconclusive results when there is little intraregional variation in raw clay materials. The microprobe also has qualities that make it exceedingly useful in the study of ceramic temper (DeAtley et al. 1982; Spurr 1993). Fired ceramics have the physical properties of metamorphic rock, making the separation of temper and paste extremely difficult (Williams 1983:302). Simply powdering the specimen before analysis produces a combined composition of the paste and temper, which is not useful for characterizing either. The ability to focus the electron microprobe beam on a very small area and determine the composition of a specific point makes it an ideal technique for ceramic temper analysis. The utility of any compositional analysis technique for answering a research question can be evaluated using three criteria-sensitivity, precision, and accuracy-as well as more mundane considerations such as speed, expense, necessary sample size, and whether the sample is destroyed during analysis. Sensitivity refers to the concentration of an element required for its detection. Elements in concentrations above 1 weight percent (wt %) are considered major elements, those between 1 weight percent and 0.1 weight percent are considered minor elements, and those that contribute less than 0.1 weight percent are referred to as trace elements (Ragland 1989:5). Due to the nature of X-ray production in the microprobe, there is a constant low-level background emission of X-rays from any sample, which hinders the detection of elements present in very small concentrations. The microprobe can detect V.4.2 |