| OCR Text |
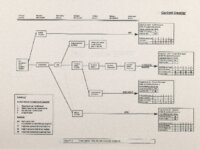
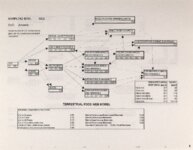
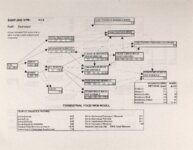
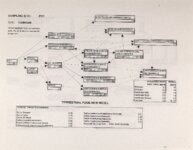
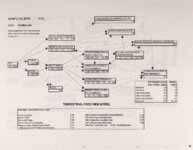
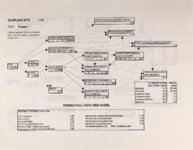
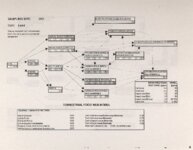
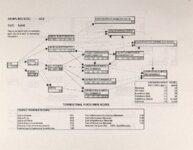
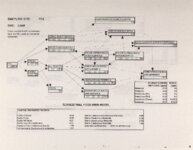
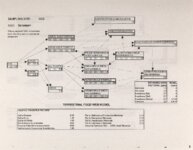
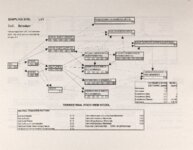
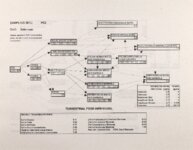
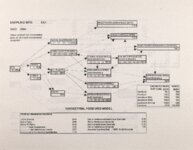
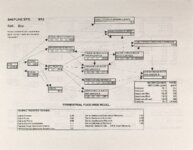
Show Ecological Risk Assessment Northern Oquirrh Mountains Appendix 2 derived are give", below the toxicity threshold table, although the actual values are not presented here. It was deemed sufficient to ascertain that chronic values were lower than acute values. 2.3 Tr:ophic Transfer Factors The potential for the chemicals of concern to accumulate in the terrestrial food chain is determined by the ratio of the chemical concentration in animal tissue to the concentration in its food. If this ratio (known as the trophic transfer factor) is greater than one, then the chemical is considered a bioaccumulator (i.e., that is, the concentration in the consumer animal's tissues is greater than the concentration in its food). Animals at the top of a food web are more at risk from a bioaccumulative compound than are lower trophic level animals. Trophic transfer factors can be calculated from whole body residue data or for each organ (e.g., liver, kidney, bone) separately. Soil-dwelling invertebrates can accumulate chemicals directly from the soil, so a soil-to-invertebrate transfer factor can also be calculated. Trophic transfer factors for the CoC were taken directly from the literature, when available. However, these data are sparse so transfer factors were calculated from information provided in research reports. For example, many papers reported the amount of chemical in feed and the amount of ct'lemical in the tissues of the species of interest, but did not calculate the ratio of tissue concentration: feed concentration. Therefore, these data were extracted from the reports and the calculations were made. Trophic transfer factors were then grouped by trophic level (e.g., data from all herbivores were grouped together, carnivore data were grouped, etc.). The mean and 95% confidence intervals of these trophic transfer values for each trophic level were calculated and reported in the tables at the end of each description of CoC toxicity values. The animal species and reference papers that were used in these calculations are also identified in the tables. 2.4 Arsenic Arsenic is a relatively common element in soil and water and is present in trace amounts in nearly all plant and animal tissues (Eisler, 1988). Arsenicals have been used in medicine for over 2,000 years and have been preferred homicidal and suicidal agents for nearly as long. This illustrates that animals are able to tolerate small amounts of As, even over extended periods, but larger doses can be acutely fatal. The NRC (1980) suggests that feed additives of As promotes growth in rats, pigs, and other animals. Chronic high exposure is associated with liver, kidney, and heart damage; hearing loss; brainwave abnormalities; rough hair coat; and bright red Chronic As poisonlnq is rarely seen in most domestic mucosa (Eisler, 1988; Osweiler, 1985). animals but is well documented in humans (NRC, 1980; Osweiler, 1985). Arsenic occurs in the environment in several states and is readily volatilized to the atmosphere when in its reduced form. The toxicity of the various forms is as follows (from greatest to least toxicity): arsines> inorganic arsenites > organic trivalent compounds (arsenoxides) > inorganic arsenates > organic pentavalent compounds > arsonium compounds > elemental As (Eisler, 1988). Methylation of As (unlike methylation of mercury) greatly reduces toxicity and is a detoxification process both in aquatic environments and within animals (Eisler, 1988). While As can accumulate in water, there is no evidence of biomagnification in the aquatic food chain (Eisler, 1988). The trivalent form is much more soluble than others, thereby increasing its potential for toxic effects (Osweiler, 1985). ecological planning and toxicology, inc. 5 |