| OCR Text |
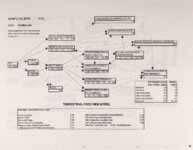
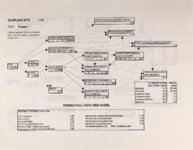
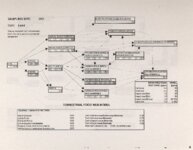
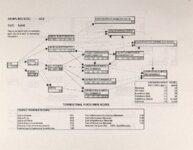
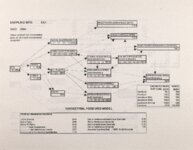
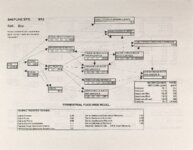
Show - Ecological Risk Assessment North Oquirrh Mountains Appendix 2 weight was given to data on wildlife species when determining a No Observable Adverse Effect Level (NOAEL) or Lowest Observable Adverse Effect Level (LOAEL) value. Both laboratory and field studies were used as appropriate. Each article was reviewed to ascertain the scientific quality of the study. A study was not used if no control or reference groups or if it was a case report on one or two animals. Sample size sufficiency was an important criterion, but allowance was made for smaller sample sizes for large animals (cattle, horses, sheep) or hard-to-keep wildlife (e.g., woodchucks). The appropriateness of statistical tests was evaluated if the conclusions from the report were to be used, but data from ·tables were used for determinations of trophic transfer factors regardless of statistical manipulations. Procedures for analytical chemistry generally are difficult to evaluate from scientific reports in peer-reviewed journals as methods sections are necessarily kept short. Methods were considered adequate if the type of analysis was reported (e.g., atomic adsorption spectrophotometry) and a reference given for details of methodology. Quality control and assurance measures (e.g., duplicates, splits, spikes, etc.) were rarely reported in the scientific literature on animal toxicology until the past few years so it was difficult to use this as a criterion for acceptance of a report. there were No Observable Adverse Effects Levels were determined by ciling the highest value reported in any of the acceptable studies that did not cause an adverse response and was lower than the lowest reported value that did cause a response. Maximum tolerable concentration (MTC) is used synonymously with NOAEL. The Lowest Observable Adverse Effect value (LOAEL) was determined as the lowest reported concentration that caused an adverse effect. A change in a blood biochemical decreased ALAD was not considered an single parameter (e.g., activity) adverse effect as the relationship as a fitness correlate has not been established. Multiple biochemical changes such as decreased thyroid hormone and white blood cell count with a concomitant increase in eorticosterone indicative of immunosuppression were considered an adverse effect as they reflect a syndrome that can reasonably be expected to reduce fitness. Effects threshold (ET) is used synonymously with LOAEL. Animals were grouped by trophic level within class for determination of NOAEL and LOAE:L values. The sample table shown here is included at the end of the discussion for each element with the appropriate information summaries (See Tables 13 through 26). All feed or tissue concentrations are reported as dry weight (d.w.) unless otherwise specified. 2.2 Acute Toxicity It is important to verify that the chronic toxicity thresholds are not higher than any known acute toxicity values. It is highly unlikely that this would be the case, as chronic values with continued (or repeated) exposure are almost always lower than a single exposure acute value. However, due to the variety of species involved, it was deemed important to do such a comparison to be certain that a particularty sensitive species was not missed. A quick search of summary li!terature augmented by some papers from the primary literature was conducted. Because acute toxicity studies report doses that were administered (Le., mg/kg-body weight), the chronic toxicity threshold values for each trophic level were converted from mg/kg feed to mg/kg-body weight/day for comparative purposes. [Note that it was considered inappropriate to change acute dosing rates used in oral gavage or injection studies to a mg/kg feed basis.] The allometric equations of Nagy (1987) were used to estimate the amount of food consumed per day for use in the conversion of feed concentrations to dose. There were no instances where the acute toxic dose was lower than the chronic feeding value (converted to a dose basis). For each CoC, the references from which acute toxicity values were 4 ecological planning and toxicology, inc. |