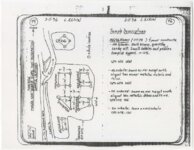
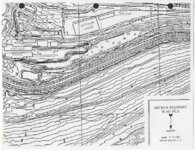
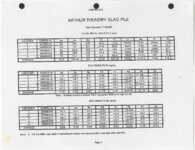
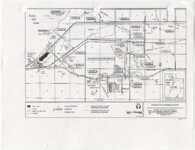
| OCR Text |
Show The Silver Building processed anode slimes from the Tankhouse by thickening them with a filtered decant solution. The thickened slimes were pressure leached with sulfuric acid and oxygen. This removed over 99% of the copper content. The residue was filtered, washed, and air dried in a filter press. Elemental selenium, silver, and gold were produced at the Refinery. Refinery waste effluent solutions were pumped to the WWTP for neutralization and heavy metals removal. The Electrolyte Purification Building was used to maintain the electrolyte solution, control copper content and soluble impurities such as arsenic, antimony, and bismuth, and control organics by electrowinning in cells containing insoluble lead anodes. The Chemical and Physical Laboratory was used as an assay laboratory to support Kennecott operations and quality .control. The building was approximately 50 feet by 100 feet with a main floor and partial basement. The contaminants associated with the Lab were found in the table hoods and dust. The Lead Shop was used to construct and mold the liners for electrowinning cells used at the Electrolyte Purification Building. The building is a small structure that has been cleaned and is currently being used as a shop with an adjoining lunch room. C.3.4 Wastewater Treatment Plant Area The Kennecott WWTP is located adjacent to the intersection of State Highways 201 (2100 South) and 202, north of the Refinery. The primary function of the WWTP was to neutralize acid plant blowdown from the Smelter and bleed electrolyte from refinery operations. The WWTP also received and treated in-plant sewage and clarified other process waters from the Smelter, Refinery, Power Plant, and the North Concentrator facilities. The sludge and wastewater have been sequentially discharged into five ponds, designated Ponds A, B, C, C-Extension, and D, all located within 1-1/2 miles of the WWTP. Treatment processes at the WWTP have been modified several times during past operations through the addition of lime, ferric chloride, or both. These modifications resulted in concentration variations of both total and leachable metals in the sludge. From start-up in 1974 through 1978, ferric chloride was added to the wastewater influent to stabilize some of the heavy metals. The addition of ferric chloride was discontinued in 1982 due to operational difficulties, but was resumed in August 1991 to reduce the concentration and leachability of arsenic in the effluent. During the period 1983 to 1989, the WWTP was modified to use a high concentration lime process. The WWTP was completely shut down between 1985 and 1987. In mid 1989, the neutralization process was changed by reducing the pH from 12 to 10 to provide better control of the pH of the final effluent at the discharge location. The addition of ferric chloride resumed in August 1991 to reduce the concentration of arsenic in the effluent and to reduce the leachability of arsenic from the sludge. Most of this low-lime sludge was stored in Pond D, b1;1t some ferric chloride-treated, low-lime sludge was plac_ed in Ponds Band C. The WWTP treated approximately 3000 gallons per minute (gpm) of which approximately 1250 gpm came from the Smelter, 1300 gpm from the Power Plant, 300 gpm from the Refinery, and 150 gpm Appendix C* 14 June 25, 1997 Rev.I |