| OCR Text |
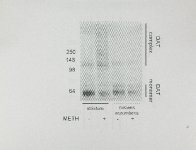
Show 59 2005). While these methods have yielded important information, they are limited in that they often utilize which may not over-expressed, modified, or truncated target proteins represent the protein in vivo. Thus, there remains a need to develop additional methodologies to identify PPls and their role in human health and disease. The present report describes a novel method to identify PPls involving the dopamine (DA) transporter (OAT). The OAT is extracellular DA from the transports synaptic a transmembrane cleft into the terminating and regulating dopaminergic signaling. protein that neuron thereby A rat striatal synaptosomal subcellular fraction was determined ex by measuring DA uptake in synaptosomal preparations. number of OAT-interacting proteins vivo selected for study have because been OAT function identified is often A including protein phosphatase 2A (Bauman et aI., 2000), a-synuclein (Lee et aI., 2001), PICK1 (Torres et aI., 2001), Hic-5 (Carneiro et al., 2002), syntaxin 1 A (Lee et aI., 2004), receptor for activated C kinase 1 (Lee et aI., 2004), protein kinase C (PKC; Johnson et aI., 2005), Ca2+/calmodulin-dependent protein kinase II (CaMKII; Fog et aI., 2006), 02 receptor (Lee et aI., 2007), G protein-coupled receptor 37 (Marazziti et aI., 2007), and synaptogyrin-3 (Egana et aI., 2009). These interactions contribute to the function and regulation of the OAT (for review, see Torres, 2006). The present study not only confirmed some of these interactions in a synaptosomal preparation, but also identified a novel potential OAT PPI involving N-ethylmaleimide (NEM) sensitive factor (NSF). The methodology described herein can be readily adapted to assess interactions involving other |