| OCR Text |
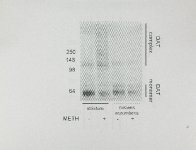
Show 8 membrane and in intracellular compartments, such as the endoplasmic reticulum Using cysteine cross-linking reagents, the and endosomes (Sorkina et aI., 20.03). OAT forms a tetramer which is localized in the plasma membrane (Hastrup at aI., Preliminary and on-going studies, using 2003). subcellular differentiation techniques, have suggested that OAT complexes at 24 h after treatment are not detected in fraction enriched with a early endosomal markers and are only detected in a fraction enriched with plasma membrane markers (data not shown). However, localization the of OAT complexes has not been conclusively determined. As stated above, numerous factors contribute to METH toxicity. The recent finding the timing and sequence of many of these factors is unclear. that multiple, high-dose formation as METH administrations also However, cause OAT complex early as 12 h after treatment (Baucum et aI., 2004) is significant because other drugs and OAT dosing paradigms which affect activity (Le., multiple MOMA administrations or a single, high-dose METH administration) do not cause OAT complex formation. Thus, if OAT complexes are associated with METH-toxicity, there exists a biomarker for drug-mediated dopaminergic toxicity which manifests hours after the insult and days to weeks dopaminergic deficits become evident. a represent a novel marker to From before scientific standpoint, this Gould rapidly identify agents which dopaminergic toxicity and to understand the toxicology of METH. as METH toxicity resembles some persistent aspects of may cause Furthermore, Parkinson's disease, understanding the functional consequences and underlying mechanisms of OAT |