| OCR Text |
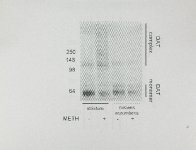
Show 6 complex formation, as exposure to the reducing agent, beta-mercaptoethanol, reverses this process The (Baucum et aI., 20'0'4). that al, (20'0'4) observed immunoreactivity occurs concurrently Baucum et formation, and there is monomer OAT METH-induced of composition no OAT a with complexes decrease in band and approximately 120 kDa not DAT METH-induced immunoreactivity detected is known. monomer OAT complex between the OAT (Baucum et al., 200'4). There are multiple possibilities that may contribute to this phenomenon, including (1) the DAT binding to large (2!: oxidative adducts, -60' kOa) proteins, (2) multiple phosphorylation, glycosylation, combination of these possibilities. or modifications ubiquitination), or (i.e., (3) a Considering the approximately 60 kDa shift, between the DAT monomer and the smaller portion of the OAT complexes (-120' kDa), and the presence of DAT immunoreactivity without discrete bands larger than approximately 120' kDa, the latter possibility is strongly suqqested (Baucum et aI., 20'04). METH-induced alterations of DAT DAT complex formation protein-protein interactions. could This is also an occur through intriguing possibility because the OAT has been shown to have numerous protein-protein interactions (for review, numerous see Eriksen et aI., 20'10' and Galli, 200'6). OAT interacts with proteins including protein phosphatase 2A, a-synuclein, protein kinase C-J3, calmodulin kinase II, hic-5, and PICK1 (Bauman at al., 20'00'; Torres at aI., 20'0'1; Carneiro et aI., 200'2; Johnson et aI., 20'0'5; Wersinger and Sidhu, 20'05; Fog et aI., 200'6). Many of these interactions have been shown to have |