| OCR Text |
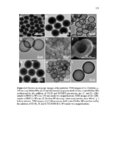
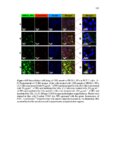
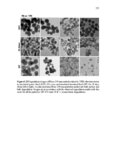
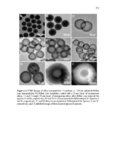
Show 273 average diameters of 190 ± 10 nm. As evident in microscopy images, highly uniform nanoparticles were synthesized. Reaction parameters that affected the physicochemical properties of the nanoparticles were concentration of precursors, how fast the precursors were added (time of injection), the amount of basic catalyst, CTAB concentration, temperature, stirring rate, and water to ethanol ratio. Scanning transmission electron microscopy (STEM) was used to evaluate approximate atomic densities of the species A and B nanoparticles. STEM images (Figure A.2) show homogeneous distribution of sulfur in the nanoparticles. Both species A and B nanoparticles had a similar distribution of sulfur. Data from X-ray photoelectron spectroscopy (XPS) were used to compare the relative composition of the two particles (Table A.1). Notice that when comparing the completed particle compositions, the relative sulfur content of particle A is larger than that of B, which is to be expected as particle A’s layers are comprised of a higher overall ratio of the disulfide-containing BTESPD. Hydrodynamic diameters of the nanoparticles were measured using dynamic light scattering (DLS) (Table A.2). The average hydrodynamic diameters of the completed A and B species particles in deionized (DI) water were: 413 ± 29, and 527 ± 61, respectively. These nanoparticles, like other spherical mesoporous silica particles, can then be easily redispersed by sonication. Zeta potential measurements were conducted at 25 °C and pH7.2 (mimicking intracellular pH) in DI water. After dispersing the nanoparticles in DI water, negative zeta potential values of around −20 mV confirmed the presence of silanol (Si– OH) groups on the surface of these nanoparticles which become deprotonated (Si–O−) in aqueous milieu (Table A.2). The A species particles tended to stay suspended for longer periods (4-6 h) than the B species (20-30 min). Nitrogen adsorption–desorption isotherm |