| OCR Text |
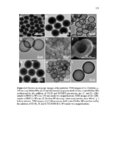
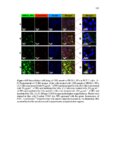
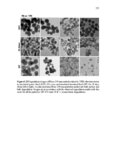
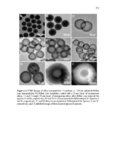
Show 107 imaging agents (e.g., iron nanocomposites) in vivo. Further studies are required to evaluate subacute toxicity, chronic toxicity, and genotoxicity of these nanoparticles both in vitro and in vivo. After determining cytotoxicity of these intact nanoparticles, toxicity of degradation products in supernatant solution was evaluated in RAW 264.7 macrophages using CCK-8 (Figure 3.23). Supernatant solutions were directly added to RAW 264.7 macrophages after being collected at each time point (0, 1, 4, 7, and 15 days). Interestingly, as demonstrated in Figure 3.23, ca. more than 85% cell viability was observed after 24 h incubation in all the time points. Ten to fifteen percent cell death may be the result of alkyl containing silicate (SiO42-) groups released from the nanoparticles. Using the CCK-8 assay, IC50 values (Table 3.4) spanned approximately from 233 ± 42 µg ml-1 to 705 ± 17 µg ml-1 for 110 Mesoporous T and 110 Mesoporous D nanoparticles, respectively. Both IC50 and fast degradation rate of 110 Mesoporous D nanoparticles suggest that this nanocarrier could be a good choice for further in vivo evaluation and its potential use for delivery of anticancer drugs to tumor tissues via enhanced permeability and retention (EPR) effect [33]. 3.3.4 In vitro cell uptake, co-localization, and release Cell uptake and intracellular co-localization of the nanoparticles were evaluated using TEM and confocal laser scanning microscopy (CLSM), respectively (Figures 3.24 and 3.25, respectively). For cell uptake, 80 µg ml-1 of the nanoparticles was added to RAW 264.7 macrophages incubated for 4 h and 24 h. Eighty µg ml-1 was selected since all the nanoparticles showed no toxicity in RAW 264.7 macrophages at this concentration after |