| OCR Text |
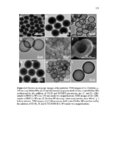
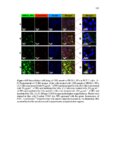
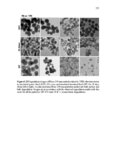
Show 259 v Disulfide-based hollow mesoporous nanoparticles started degrading from the very first hours due to thin shell and hollowness and disintegrated into smaller fragments. v Hollow mesoporous silica nanoparticles can incorporate doxorubicin as a model drug ca. 10 times higher than the same size nonhollow mesoporous counterparts. v In vitro, intracellular, and in vivo degradation results demonstrate that nanoparticle porosity and core composition as well as the pH of the medium play the predominant roles for degradation of silica nanoparticles. v Fabricated silica nanoparticles degraded very slowly in simulated gastric fluid pH 1.2 over 28 days. v Compared to other nanoparticles studied, regular mesoporous particles with an average diameter of 100 nm degraded at a faster rate at higher pH values with 100% degradation within a week in simulated intestinal fluid pH 6.5 and simulated body fluid pH 7.4. v Disulfide-based degradable particles underwent both hydrolysis and disulfide reduction degradation in the reductive environment and disintegrated into smaller fragments. However, these particles did not exhibit faster degradation rate compared to regular mesoporous nanoparticles composed of TEOS likely due to the hydrophobic nature of the precursor used in the synthesis of disulfide-containing nanoparticles. v In vivo results indicate that at day 7 postintravenous injection in CD-1 mice particle porosity and density do not have drastic effects on biodistribution. Based on these findings, it can be concluded that by fabricating hollow silica nanoparticles as well as choosing the right precursor(s), it is possible to design and develop nanoparticles with optimized loading capacity, controlled degradation profile, and reduced toxicity for utility in specific delivery applications. |