| OCR Text |
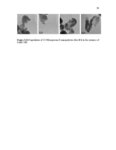
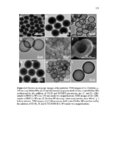
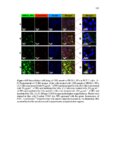
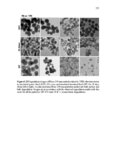
Show 66 size distributions were obtained from an adsorption branch by using the Barrett-JoynerHalenda (BJH) method. The Brunauer−Emmett−Teller (BET) specific surface areas were measured via adsorption data at P/P0= 0.05−0.20. Scanning transmission electron microscopy (STEM) images and spectra of the nanoparticles were acquired on JEOL JEM2800 (Akishima, Tokyo, Japan) scanning transmission electron microscope with dual energy dispersive X-ray spectrometer (EDS) detectors at an electron beam energy of 200 kV. Sample preparation was done by drop casting the nanoparticles on a carbon coated TEM grid. X-ray photoelectron spectroscopy (XPS) analyses of the nanoparticles were performed by Axis Ultra DLD instrument (Kratos Analytical, Manchester, UK). For analyses, the samples were mounted on a C tape and pumped overnight in the load lock before introduction into the analysis chamber. A mono Al source was employed. Survey scans were collected with a pass energy of 160 eV, step size of 1 eV, and dwell time of 200 ms. High resolution region scans were collected with a pass energy of 40 eV, 0.1 eV step size, and 400 ms dwell time. Data were processed using CASA XPS software. Thermogravimetric analyses (TGA) were performed using a TA Instruments hi-res TGA 2950 Thermogravimetric Analyzer (New Castle, DE, USA). All TGA experiments were performed under N2 atmosphere from 35 to 800 °C at a heating rate of 20 °C/min. 3.2.4 Degradation studies Degradation of all the synthesized nanoparticles (80 µg mL-1) was evaluated mimicking intracellular and extracellular GSH concentrations (8 mM and 8 µM, respectively) in DI water at 37 °C and under constant rotation at 18 RPM using ProBlot™ 100 hybridization incubator. At predetermined time points (0, 1, 4, 7, and 15 days), samples |