| OCR Text |
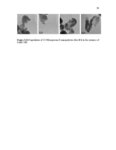
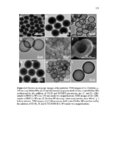
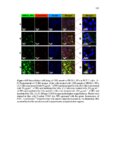
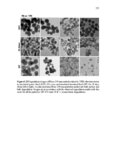
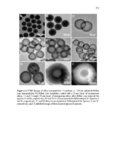
Show 247 chosen as it was shown in Figures 5.29-5.31 that particles are taken up more by this organ. As indicated in Figure 5.33B-D, particles are taken up in a group (Meso 100: Figure 5.33C) or as individual particles (Stöber 100: Figure 5.33B and Disulfide Hollow 100: Figure 5.33D). Control group treated with 0.9% normal saline is shown in Figure 5.33A. Animals were observed on a daily basis. No clinical toxicity or behavioral changes were observed during the treatment period and no significant fluctuations in body weight were seen in control and nanoparticle treated groups (Figure 5.34). Overall, the results of this study indicate that SiO2 NP porosity and composition, and the pH of the environment play the predominant roles for the degradation of SiO2 NPs. As observed, regular mesoporous particles (Meso 100 and Meso 500) exhibited faster degradation in all the studied media which could be attributed to high surface area of these particles. Higher surface area enhances nanoparticle exposure to water molecules and hydroxyl groups which can initiate the degradation process resulting in the cleavage of SiO-Si bonds. In SBF with 10 mM of GSH, the degradation of the disulfide-based degradable mesoporous particles was clearly slower than their regular mesoporous counterparts. This could possibly be attributed to the hydrophobic nature of the precursors comprising carbon chains used for the fabrication of these disulfide-containing particles. In addition, the influence of the degradation media pH is prominent as the degradation rates of all the particles considerably increased by increasing the pH especially to pH values above 4.5. In particular, the fastest degradation rates were observed at pH values close to neutral. At higher pH values, higher concentration of hydroxyl ions enhances nucleophilic attack on Si atoms leading to the formation of unstable penta-coordinated intermediates, breakage of |