| OCR Text |
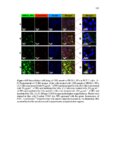
Show 126 respectively. The cells were then incubated for another 24 and 48 h, the media was aspirated, and the cells were washed twice with PBS. Cell viability was determined using CCK-8 cytotoxicity assay kit according to an established protocol and absorbance was measured at 450 nm with SpectraMax® M2 microplate reader. Assays were performed at least in triplicate. Cytotoxicity of the intact GSH-sensitive HMSiO2 NPs was also evaluated in RAW 264.7 macrophages and in NIH 3T3 fibroblasts. Cells were cultured at 37 °C in 5% CO2 in RPMI-1640 and DMEM (for RAW 264.7 macrophages and NIH 3T3, respectively) with 10% FBS and seeded onto 96-well plates with the density of 4,000 cells/well and incubated to grow for 48 h. Subsequently, the cells were washed with PBS. Fresh media containing 10% FBS was then added with varying nanoparticle concentrations ranging from 0 to 1000 µg mL-1. The cells were then incubated for another 24 and 48 h, the media was aspirated, and the cells were washed twice with PBS. Cell viability was measured using CCK-8 cytotoxicity assay kit at 450 nm. Assays were performed at least in triplicate. 4.2.6 Intracellular trafficking via confocal laser scanning microscopy (CLSM) To explore intracellular trafficking and co-localization of the HMSiO2 NPs, CLSM was used and images were obtained by Olympus FluoView™ FV1000 confocal microscope (Olympus Corporation, Shinjuku, Tokyo, Japan) at 60x magnification. FITC-labeled HMSiO2 NPs were synthesized by the following procedure: 0.021 mmol of FITC and 0.021 mmol of APTES (1:1 molar ratio) were mixed in 10 mL of absolute ethanol and the reaction was kept under stirring (400 RPM) for 2 h at room temperature. Then, 5 mL of absolute |