| OCR Text |
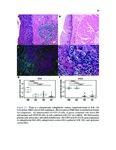
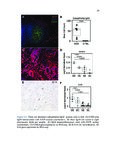
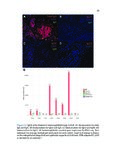
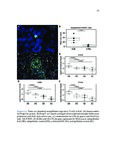
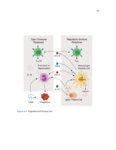
Show 70 A.1.3 Results The pulmonary infiltrate has abundant eosinophils, FoxP3+ regulatory T cells, DCSIGN+ and CD68+ macrophages, mast cells, basophils, phosphoSMAD2 expression (implying TGF-β activity), CYP27B1 expression (implying calcitriol synthesis), and many IgG4+ plasma cells. IgG4-rich granular deposits are present focally, some coating macrophages. Eosinophil degranulation is seen focally, particularly in areas of fibrin and granular immunoglobulin. RNA studies confirm that EGPA lung has abundant IGHG4 (increased 1,885-fold relative to controls, p≤2x10−15 ) and, to a lesser degree, other immunoglobulin transcripts. Statistically significant increases, each ≥8-fold greater in EGPA compared to controls, are also seen for SIGLEC8 (for eosinophils and mast cells), FOXP3, IL13, CCL18, CCL13, B cell markers, CYP27B1 (which makes calcitriol, active vitamin D) and the alternatively activated macrophage marker CD209 (DC-SIGN). A.1.4 Conclusions EGPA has both type 2 and immunoregulatory features. We propose a novel mechanism explaining this hybrid immune response. A.2 Introduction EGPA (eosinophilic granulomatosis with polyangiitis) is a rare systemic disease characterized by multiple phases - asthma, often severe and adult onset, then peripheral blood and tissue eosinophilia [1]. Multiple organs can be involved; lung eosinophil infiltrates and sometimes pleural effusions are common. Generally years later, a life threatening systemic vasculitis develops. The pathologic findings in EGPA have not been carefully studied due to the rarity of EGPA and because biopsies are not required for diagnosis and are not done. Findings in early phases of EGPA likely differ from those in autopsies of untreated patients as initially studied [1]. Vasculitis with fibrinoid necrosis, once a defining feature, is absent in early EGPA. While EGPA usually responds to nonspecific immunosuppressive treatments such as systemic glucocorticosteroids, sometimes with cyclophosphamide, the success of more targeted therapies are indicative of the underlying mechanism of disease. B-cell-depleting (rituximab, anti-CD20) and eosinophil-depleting (anti-IL5, mepolizumab) therapies are |