| OCR Text |
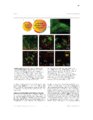
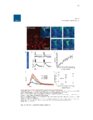
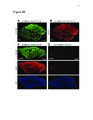
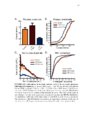
Show 99 mouse to monitor astrocytic process termini and electrophysiology to record neuronal activity, neuronal-astrocytic interactions in the TMEV-infected brain could be interrogated. Conclusion This work will has already begun to impact the glia field by offering improved approaches to observing calcium activity in glia and neurons in the rat or mouse brain (Gee et al., 2014, 2015; Pozner et al., 2015). In the immediate future, novel characterization of reactive astrocytic alterations in different models of neuropathology will be performed. With the assistance of continued fast pace of technology development for recording of cell population dynamics, it is conceivable that new treatments for many currently misunderstood neurological disorders are just on the horizon. References Akerboom, J., Chen, T.-W., Wardill, T.J., Tian, L., Marvin, J.S., Mutlu, S., Calderón, N.C., Esposti, F., Borghuis, B.G., Sun, X.R., et al. (2012). Optimization of a GCaMP calcium indicator for neural activity imaging. J. Neurosci. Off. J. Soc. Neurosci. 32, 13819-13840. Araque, A., Parpura, V., Sanzgiri, R.P., and Haydon, P.G. (1999). Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci. 22, 208-215. Beattie, E.C., Stellwagen, D., Morishita, W., Bresnahan, J.C., Ha, B.K., Zastrow, M.V., Beattie, M.S., and Malenka, R.C. (2002). Control of synaptic strength by glial TNFα. Science 295, 2282-2285. Beers, D.R., Henkel, J.S., Schaefer, D.C., Rose, J.W., and Stroop, W.G. (1993). Neuropathology of herpes simplex virus encephalitis in a rat seizure model. J. Neuropathol. Exp. Neurol. 52, 241-252. Bernardinelli, Y., Randall, J., Janett, E., Nikonenko, I., König, S., Jones, E.V., Flores, C.E., Murai, K.K., Bochet, C.G., Holtmaat, A., et al. (2014). Activity-dependent structural plasticity of perisynaptic astrocytic domains promotes excitatory synapse stability. Curr. Biol. CB 24, 1679-1688. |