| OCR Text |
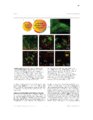
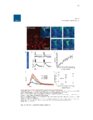
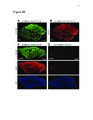
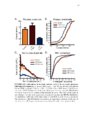
Show 4 indicators have evolved quickly and become the first choice for optically monitoring astrocyte activity (Nimmerjahn and Bergles , 2015). Since the 1980s, there have been many iterations of synthetic calcium dyes, based on the EGTA analog, BAPTA (Gee et al., 2000; Grynkiewicz et al., 1985; Tsien, 1980). Unfortunately, synthetic dyes are associated with many drawbacks, including major limitations on what types of tissue for which they can be useful (Peterlin et al., 2000; Reeves et al., 2011). Because only young tissue is able to take up dye via the bulk loading technique, most early astrocyte studies were performed in relatively immature tissue or in culture (Cornell-Bell et al., 1990; Kim et al., 1994). An additional complication with synthetic dyes is that they do not readily diffuse into astrocytic processes where interesting neuronal-astrocytic interactions occur, unless invasive techniques such as pipette loading are used (Reeves et al., 2011). These limitations motivated the development of genetically encoded calcium indicators (GECIs). Although the first calcium indicator, the naturally occurring jellyfish protein aequorin, was genetically encoded (Shimomura et al., 1962), it was not until green fluorescent protein (GFP)-based probes became available that GECIs could start to compete with properties of synthetic dyes. Early GECIs suffered from performance issues at physiological temperature and pH (Miyawaki et al., 1997; Nakai et al., 2001). Iterations of the GCaMP family have offered indicators without these issues but with similar speed and signal-to-noise ratio relative to synthetic dyes while also permitting expression in mature tissue and labeling of cellular processes (Akerboom et al., 2012; Chen et al., 2013; Shigetomi et al., 2013; Tian et al., 2009). The kinetics and sensitivity of GECIs continue to improve at a rapid pace (Nagai et al., 2014; Peron et al., 2015; Rose et al., 2014). Calcium imaging has already played a crucial role in interrogating astrocyte function. The revelation that cultured astrocytes respond to extracellular glutamate with an |