| OCR Text |
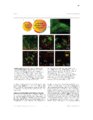
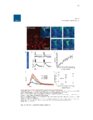
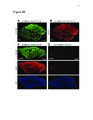
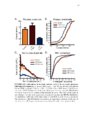
Show 62 between process ROIs decreased over time as well, supporting the qualitative observation that events in LPS-exposed astrocytes progressively decreased in size. Future work will be focused on identifying specific calcium source contributions to intrinsic activity in each subcellular compartment in the healthy and inflamed brain. This work has generated interesting questions regarding astrocyte function in the diseased brain which will facilitate discovery of novel drug targets for disease modifying therapies. Introduction Astrocytes are the most common nonneuronal neural cell type in the brain. In response to a neurological insult, including trauma, infection, or seizures, astrocytes convert to a reactive state defined by a variety of dramatic morphological and molecular alterations. It is not understood how these alterations affect astrocyte function and whether they play a net beneficial or detrimental role in disease pathogenesis and recovery. The recent development of fluorescent probes for monitoring cellular activity has permitted direct observation of various types of astrocytic dynamics in adult and pathological tissue. In particular, genetically encoded calcium indicators (GECIs), such as the GFP-based GCaMP family, have become essential for observing dynamics in nonelectrically excitable cells, including astrocytes. GECIs have enabled direct monitoring of alterations in astrocytic calcium dynamics in response to various pathological processes. In this report, we describe changes in intrinsic calcium activity in both astrocytic somas and fine processes during the acute phase of endotoxemia-induced reactive astrocytosis. Lipopolysaccharide (LPS; also called endotoxin) is a component of the gramnegative bacterial cell wall that activates the innate immune system (Beutler, 2000) via Toll Like Receptor 4 (TLR4) signaling. TLR4 is expressed by macrophages and monocytes |