| OCR Text |
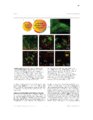
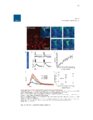
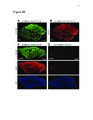
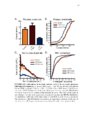
Show 97 Recent evidence supports a role for astrocytes in maintaining synaptic strength through uptake, metabolism and release of gliotransmitters such as ATP, glutamate and Dserine (Beattie et al., 2002; Butt, 2011; Jourdain et al., 2007; Pascual et al., 2005; Stellwagen and Malenka, 2006; Trasande and Ramirez, 2007). In particular, astrocyte glutamate release has been proposed to strengthen synapses (Jourdain et al., 2007; Santello and Volterra, 2009) and prolong action potentials (Sasaki et al., 2011). Therefore, it is plausible that altered astrocyte glutamate release could cause synaptic hyperexcitable (Savin et al., 2009). If this occurs in a large number of local synapses, a hyperexcitable region, or epileptogenic focus, could emerge and contribute to seizure generation. Therefore, there is reason to believe that astrocyte glutamate release may contribute to epileptogenesis. In epileptic patients, reactive astrocytosis is often observed in the HC (Thom et al., 2009), which is also commonly determined to be the focus of epileptiform activity (Oliveira et al., 2011). It has been demonstrated that reactive astrocytes exhibit altered glutamate release in a model of Alzheimer's disease (Rossi et al., 2005). However, astrocytic functional alterations in the epileptic brain are not well understood (Seifert et al., 2010; Wetherington et al., 2008). It has been demonstrated that a proinflammatory cytokine, TNF-α, is required for synaptic scaling by permitting glutamate release from astrocytes onto neurons (Santello and Volterra, 2009). This raises the question if high levels of TNF-α in the brain, such as what is observed during an infection of the brain, could promote synaptic potentiation. This idea is especially important in viral-induced epilepsy which is characterized by a robust immune response in the brain that occurs before the onset of seizures (Getts et al., 2008). It is unclear how the immune response and seizure development are linked (Vezzani et al., 2011). |