| OCR Text |
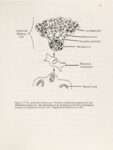
Show CHAPTER 4 THE INCORPORATION OF PHENCYCLIDINE INTO HAIR: A COMPARISON WITH MELANIN CONCENTRATION There are two types of melanin: eumelanin and pheomelanin. Eumelanin is a brown-black polymer of dihydroxyindole subunits synthesized from the precursor amino acid tyrosine. Eumelanin has a quinone-like structure and is highly anionic. Pheomelanin is a reddish-yellow pigment also synthesized from the precursor tyrosine. However, the incorporation of sulfhydryl groups from cysteine or glutathione result in benzothiazine derivatives which polymerize to pheomelanin (Figure 1.3). Both eumelanin and pheomelanin can be present in hair and the combination of these two types of melanin in mature hair dictate the resulting color (Table 1.1). Much of the published research describing melanin as an adsorbent of drug molecules has focused on eumelanin (98,103,105,107,108,110,136,137). Sepia melanin, isolated from squid ink sacs, is used in a number of in vitro studies and has eumelanin as its primary component. Melanin isolated from the uveal tracts of bovine eyes is also predominantly eumelanin (111, 112, 138). The use of these sources of melanin has provided a great deal of information regarding the binding of drugs to eumelanin, but no studies have focused specifically on the binding of drugs to pheomelanin. The lack of pheomelanin data is likely due to the fact that there is no commercially available source of pheomelanin in spite of its published synthesis (138). It is reasonable to theorize that drugs might bind to pheomelanin. The free carboxyl, and amine groups |