| OCR Text |
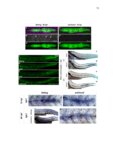
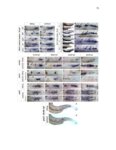
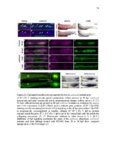
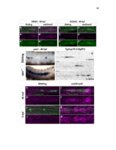
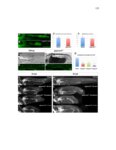
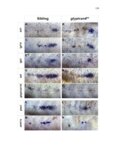
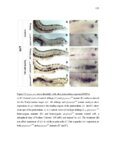
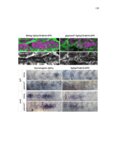
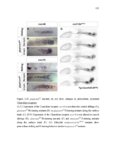
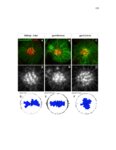
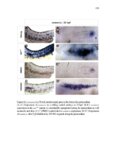
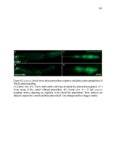
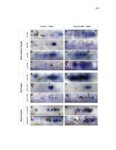
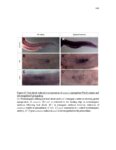
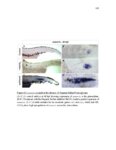
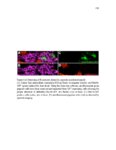
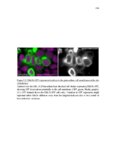
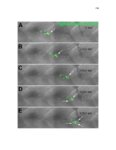
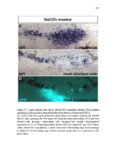
Show 47 H). In contrast, cxcr7b expression is only downregulated at 44 hpf and disappears by 50 hpf (Figure 2.8I-P). The loss of cxcr7b causes a decrease in migration speed. Hence, in contrast to WT prim, mutant prim is delayed (Figure 2.2T). Prim migration is also influenced by Cxcl12a expression in a subset of Engrailedpositive muscle pioneer cells (MPCs) and medial fast muscle fibers (MFFs) along the myoseptum that form a track along which the prim migrates (Dambly-Chaudiere et al., 2007; David et al., 2002; Li et al., 2004; Valentin et al., 2007). These muscle cells form in response to Shh secreted by the notochord, whereas their development is restricted to the midline by Bmp repressing signals from the somites (Maurya et al., 2011). Cxcl12a is upregulated along the myoseptum in extl3/ext2 mutants (Figures 2.8Q-X) in which the number of cxcl12a secreting MPCs does not change but the MFFs are increased in response to higher Shh signaling (Figure 2.8Y-Z and Figure 2.9A-D’). Interestingly, extl3/ext2 mutants show upregulation of cxcr4b and cxcl12a at 32 hpf (Figure 2.8A-B and Figure 2.8Q-R) yet the prim migrates for 12 more hours (Figure 2.8E-8F and Figure 2.8U-V) and only slows down once cxcr7b is downregulated at 44 hpf (Figure 2.8M-N). This delayed phenotype implies that simultaneous upregulation of cxcr4b and cxcl12a in the presence of cxcr7b allows for the temporary maintenance of the Cxcl12a gradient required for prim migration. As Wnt inhibits the expression of cxcr7b in the primordium, the changes in cxcr7b expression correlate with Wnt signaling expansion (Figure 24Q-Q’ and 2.9G-J;) (Aman and Piotrowski, 2008). The reduction of cxcr7b expression marks the time point when prim cells begin to tumble and stall suggesting that the loss of directional migration is likely caused by primordiumautonomous defects in chemokine receptor expression. |