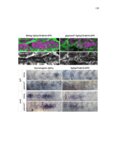
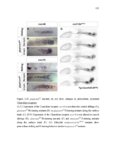
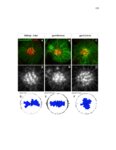
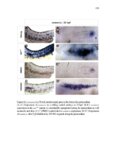
| OCR Text |
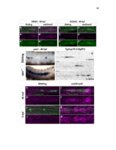
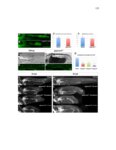
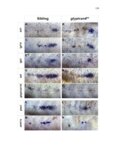
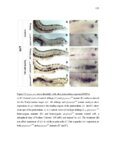
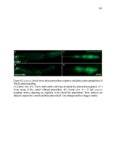
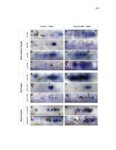
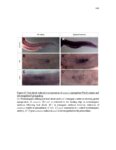
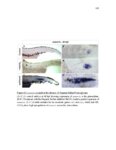
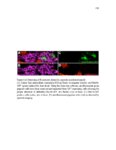
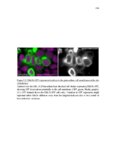
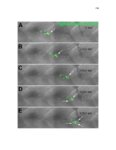
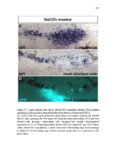
Show 36 the Fgf inhibitor sef and the Wnt inhibitor dkk1b, respectively. This ensures the segregation of the Wnt and Fgf activation domains to adjacent territories (Aman and Piotrowski, 2008). However, the mechanisms regulating ligand distribution and its effects on activation of signaling cascades to coordinate cell migration remain to be elucidated. Extracellular matrix proteins, such as Heparan Sulfate Proteoglycans (HSPGs) bind and regulate the activity of signaling molecules (Sarrazin et al., 2011). HSPGs possess chains of the sulfated glycosaminoglycan heparan sulfate (HS) that bind signaling molecules in the extracellular matrix. Previous studies suggest three main HSPGs functions. First, they act as coreceptors for the Wnt, FGF, Hh and BMP pathways (Kreuger et al., 2004; Lin and Perrimon, 2000; Shiau et al., 2010; Yan and Lin, 2007). Second, HSPGs alter the ability of signaling molecules to move from cell to cell (Yan and Lin, 2009; Yu et al., 2009). Third, HSPGs can be cleaved or shed from the cell membrane, changing ligand concentration and availability to adjacent cells (Giraldez et al., 2002; Manon-Jensen et al., 2010). Thus, HSPGs are important for signaling activation and potentiation of morphogen gradients. Disruption of HSPGs leads to defects in gastrulation, convergent extension and axon sorting (Clement et al., 2008; Lee et al., 2004; Poulain and Chien, 2013; Topczewski et al., 2001). However, the mechanisms through which HSPGs regulate signaling pathways or how HSPGs alterations may result in developmental abnormalities are not well defined. The Exostosin (EXT) family of glycosyltransferases synthesizes HS chains (Busse et al., 2007). Mutations in human EXT genes cause Multiple Hereditary Osteochondroma (MHO), an inherited skeletal disorder, and are associated with leukemia, breast, liver and colorectal cancer (Busse-Wicher et al., 2013). EXT genes are |