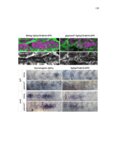
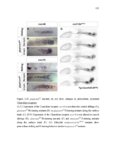
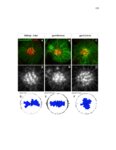
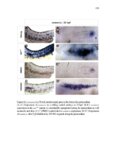
| OCR Text |
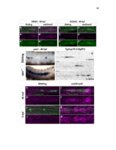
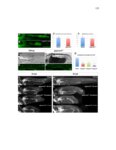
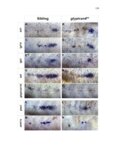
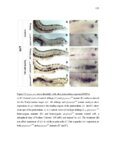
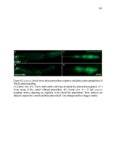
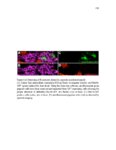
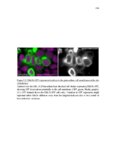
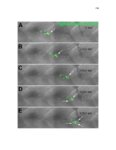
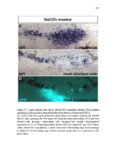
Show 10 hence manipulations that ectopically activate Wnt/β-catenin lead to complete downregulation of cxcr7b, and more importantly, failure in primordium migration . Recently, using a transgenic line expressing Cxcl12a-GFP it was confirmed that this ligand is expressed uniformly across the myoseptum. However, by using a Cxcl12a-dependent signaling sensor line which uses as a read out the internalization of Cxcl12a, it was elegantly confirmed that Cxcl12a signals only through Cxcr4b, as this receptor will internalize the ligand upon interaction on the cell membrane. However, and as previously hypothesized, Cxcr7b sequesters Cxcl12a and retains it at the cell membrane in back of the primordium. This sequestration generates an internal gradient that the primordium can sense and allow for migration (Dambly-Chaudiere et al., 2007; Dona et al., 2013; Venkiteswaran et al., 2013). Heparan Sulfate Proteoglycans Heparan Sulfate Proteoglycans (HSPGs) encompass a large group of glycoproteins that can be either secreted (Perlecans), embedded in (Syndecans) or GPIanchored (Glypicans) to the cell membrane (Figure 1.2). All of them have a core protein to which highly charged Heparan Sulfate glucosaminoglycan (HS-GAG) sugar chains are covalently attached, post-translationally (Bernfield et al., 1999; Esko and Selleck, 2002). Additionally, specialized enzymes, such as sulfotransferases, sulfatases and epimerases modify these HS-GAGs sugar chains with a series of moieties that make every HSPG unique and enable them to attract and interact with different molecules in a tissue- and time-dependent manner. The sugar chain moieties in HS-GAGs facilitate their binding to molecules in the extracellular matrix that includes Wnt, Fgf, BMP and Hedgehog (Hh) |