| OCR Text |
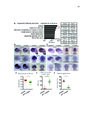
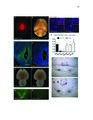
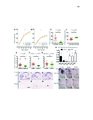
Show 69 3.7A, B), identified only 2 affected protein-coding genes with an AdjP < 0.1: Pmch and Tacr3 (Table 3.4). We confirmed reduced mRNA expression of Pmch and Tacr3, as well as Cartpt, all of which are coexpressed in neurons of the lateral hypothalamus (Brischoux et al., 2001) (Figure 3.7H, 3.7O-T, 3.6K), consistent with loss of these neurons in Lef1CKO mice. Decreased body weight observed after ablating Pmch+ neurons (Alon and Friedman, 2006; Whiddon and Palmiter, 2013) may therefore be related to a role for these cells in mediating stress and anxiety, which is further supported by characterization of their inputs and activity (González et al., 2016). Orthologs of multiple Lef1-dependent genes in zebrafish are expressed with or adjacent to Lef1 in the mouse hypothalamus (Ng et al., 2009; Visel et al., 2004), but their expression was Lef1-independent in mice (Tables 3.3, 3.4). For example, reduced Corticotropin releasing hormone binding protein (Crhbp) expression (Figure 3.3C) can lead to increased anxiety and delayed body growth (Karolyi et al., 1999), but we confirmed its unaffected expression in P22 Lef1CKO mice (Figure 3.7H, 3.6K-M). In addition, we confirmed that expression of zebrafish pmch orthologs (Berman et al., 2009) does not depend on Lef1 (Figure 3.9A-D). Therefore, while the resulting circuit function is conserved, the identity of Lef1-dependent neurons differs between zebrafish and mice. Recent work suggests that Lef1-dependent neurons in zebrafish may represent an evolutionarily ancient circuitry to regulate anxiety (Mohammad et al., 2016). Indeed, we observed expression of the lef1 ortholog pan in the stage 14 Drosophila neuroendocrine pars lateralis (PL) primordia (Hartenstein, 2006) (Figure 3.10A, B, G), and a specific loss of crhbp (CG15537) expression in the PL of pan mutants at stage 16 (Van de Wetering et al., 1997) (Figure 3.10C-H). These results are consistent with a relationship between the |