| OCR Text |
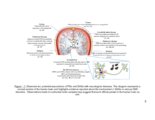
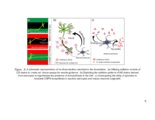
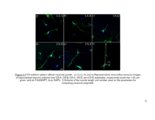
Show 181 affected region. A number of fluoro-xylosides have been found to inhibit GAG biosynthesis in CHO and endothelial cells (13, 14). In Chapter 3, it was suggested that endosulfatase secreted by reactive astrocytes might provide additional inhibitory cues to regenerating neurons. Therefore, small molecular inhibitors of GAG endosulfatases can be utilized to preserve growth factor sequestering the HS chains at the damage site. By selectively targeting one or more endosulfatases, unique 3-O-sulfatated HS motifs can be preserved to stimulate neuronal regeneration. In various neurodegenerative disorders, GAGs may enhance formation of proteaseresistant plaques; inhibiting this binding may increase the turnover of these pathological aggregates by prolonged exposure to proteases. Polysulfonated GAG mimetics have been used for this purpose and were found to be protective against amyloid fibril-induced effects (15-17). Furthermore, 4-deoxy-N-acetyl glucosamine was shown to attenuate plaque formation and improve effects of AD (18). Therefore, several permutations and combinations of small molecular inhibitors of GAG biosynthesis or desulfation could be utilized to create clinically applicable solutions for degrading or modifying pathological PGs in the CNS. Although several directions on how GAGs can be exploited to enhance CNS recovery are proposed in this work, such approaches may pose some limitations since GAGs have multifunctional roles in CNS injury and pathology. First, even though GAGs was reported to be functional units in CSPG inhibition, additional roles of the protein core of PGs cannot be excluded in other context of CNS. Next, while the use of ChABC or click-xylosides may promote short-term |