Contents | 42 of 230
Page 42
| Title | The role of glycosaminoglycan motifs in regulating neuronal growth, guidance and inhibition |
| Publication Type | dissertation |
| School or College | College of Engineering |
| Department | Biomedical Engineering |
| Author | Swarup, Vimal Paritosh |
| Date | 2014 |
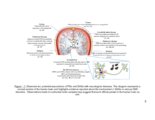
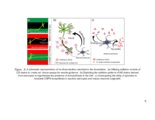
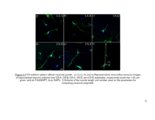
| Description | This work investigates the structure-function relationship of glycosaminoglycans (GAGs) with respect to neuronal outgrowth. Chondroitin sulfate (CS) and Heparan sulfate (HS) are the two most important kinds of GAGs found in the CNS. CS proteoglycans (CSPGs) are overexpressed in the central nervous system (CNS) after injury and form the inhibitory barriers for regenerating neurons, whereas HSPGs mediate essential growth factor-receptor interactions in the CNS. The first of part of my research explored the impact of CS sulfation patterns on growth and pathfinding of the hippocampal neurons. Specific CS variants: CS-A, CS-B, and CS-E attracted neurites, whereas CS-C was avoided by the hippocampal neurons. The differential response of neurons was exploited to create binary patterns of surface-immobilized CS chains to enhance both growth and guidance of neurons. In the second part, the importance of the GAG-sulfation pattern was further demonstrated by analyzing astrocytes HS. The astrocytes synthesized HSPGs containing a very distinct sulfation profile. The trisulfated residues containing NS2S6S sulfation were significantly lower than the disulfated residues containing NS2S sulfation. It was hence hypothesized that the secretion of endosulfatases in astrocytes condition media was responsible for conversion of the NS2S6S residues into NS2S residues. Substrate specificity for human isoform of the enzyme was also performed. It was confirmed that astrocytes secreted endosulfatase enzymes into the condition media. These results suggest the possibility of an additional mechanism through which astrocytes mediate neuronal inhibition. By removing specific HS domains that interact with growth factors, the scar extracellular matrix (ECM) could hinder the regeneration process in neurons. In the final part of the study, an approach to reduce the inhibitory GAGs secreted by astrocytes was explored. Click-xylosides reduced the amount of CS chains attached to core protein by competing with endogenous xylose residues. Upon treatment with condition media, neuronal growth was directly related to the CSPG content of the media. By using bis-xylosides or chemically conjugated xylosides-primed CS chains, it was demonstrated that multivalency is essential to impart inhibitory functions to CS chains. Overall, this part of the study explores a therapeutic strategy of reducing neuronal inhibition by exploiting click-xylosides to modulate CSPG secretion in the scar ECM. |
| Type | Text |
| Publisher | University of Utah |
| Subject | Chondroitin sulfate; Glycosaminoglycan; Heparan sulfate; Hippocampal neurons; Microcontact printing; Proteoglycan |
| Dissertation Name | Doctor of Philosophy |
| Language | eng |
| Rights Management | ©Vimal Paritosh Swarup |
| Format | application/pdf |
| Format Medium | application/pdf |
| ARK | ark:/87278/s65q922f |
| DOI | https://doi.org/doi:10.26053/0H-QMWT-WC00 |
| Setname | ir_etd |
| ID | 1353912 |
| Reference URL | https://collections.lib.utah.edu/ark:/87278/s65q922f |
Page Metadata
| Title | Page 42 |
| Format | application/pdf |
| Setname | ir_etd |
| ID | 1353954 |
| OCR Text | Show |
| Reference URL | https://collections.lib.utah.edu/ark:/87278/s65q922f/1353954 |