| OCR Text |
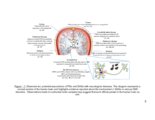
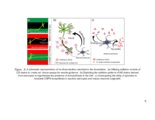
Show 179 5.2 Future Directions The findings presented in this dissertation substantiate the importance of GAGs on viability, growth, and pathfinding of hippocampal neurons. Scientific literature is replete with reports on how GAGs play a major role in CNS development and maintenance. The goal for the future will be to exploit GAGs as therapeutic agents in CNS disorders. From the reported evidence and my research findings, we understand that the fine sulfation patterns and glycosylation density of PGs influences their bimolecular interactions in the CNS. Based on these finding, multiple approaches can be device to develop therapies for neurological injuries and disorders discussed earlier. Present efforts on developing therapies for CNS injury/disorders are headed in three directions: (i) removing inhibitory CSPGs from the injury site (1); (ii) delivering neurotrophic factors for stimulating regeneration (2); and (iii) using stem cells to support neuronal regeneration (3). However, since none of these approaches have shown complete functional restoration independently, there is a need to design combinatorial therapies that modulate the complex neuro-inhibitory environment present at the scar/disease site to facilitate neuronal regeneration. Digestion of CSPGs at the scar site after injury is one of the most widely investigated therapeutic approaches for CNS injuries. Several studies have demonstrated the therapeutic efficacy of combining the digestion of CSPGs with infusions of neurotropic factors (4-7). In spite of promising results of ChABC, delivery of the active enzyme for prolonged periods has practical limitations. ChABC has been reported to be thermally unstable, decreasing its activity |