| OCR Text |
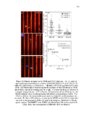
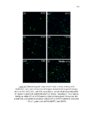
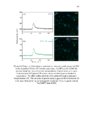
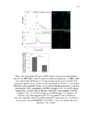
Show ! 124 residues in analysis. The 3-O-sulfation is a rare modification of HS, and is involved in crucial CNS signaling mechanisms. In this study, we verify the moieties of HS that are potential substrates of the endosulfatases (Fig 3.6). ! 3.3.5 Neither GlcA-GlcNAc6S nor GlcA-GlcNS6S is a good substrate for HSulf-1 To investigate the effect of N-acetylation/N-sulfation on HSulf-1-mediated 6-O-desulfation, we prepared a 50% N-sulfated heparosan substrate and modified it with 6-OST-3 as described in the materials section to obtain the substrate 1a shown in Fig 3.6. The disaccharide profile of the substrate 1a before and after HSulf-1 treatment is shown in Figs 3.7 (a) and (b), respectively. As evident in the figure, neither GlcA-GlcNAc6S nor GlcA-GlcNS6S was a good substrate site for the enzyme. This outcome suggests that both N-acetylation and N-sulfation do not affect the sulfatase activity. Inactivity of HSulfs on GlcAGlcNS6S has also been reported previously (14). However, in a recent study, Lamanna et al. have shown HSulfs to have some limited activity on GlcAGlcNS6S residues (27); subtle differences in the reported activity of HSulf-1 can be due to the fact that the substrates used in these studies are heterogeneous and may vary in the pattern of the sulfation of sequences flanking the target disaccharide (GlcA-GlcNS6S). Staples et al. have also reported the dependence of HSulfs on unique structural features present on HS (28). In order to confirm this conclusion, we conducted an additional experiment on substrate containing GlcA-GlcNS6S[35S] (substrate 1b) as shown in Fig 3.7 (c) and (d). However, we concluded that HSulf-1 did not show any activity on GlcA-GlcNAc6S or GlcA! |