| OCR Text |
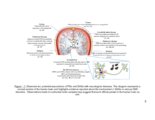
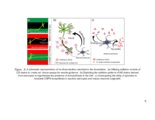
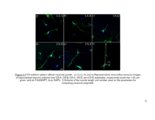
Show 16 media (CM). It is therefore likely that the endosulfatases remove 6-O-sulfates from HS present on neurons. This modulation of neuronal HSPGs could have significant manifestations of growth of neurons, as discussed in the chapter. CSPGs are well known to have varying roles in CNS injury. For example, a cell surface PG protein tyrosine phosphatase ζ (PTPζ) (also known as receptor protein tyrosine phosphatase β) is expressed on migrating neurons and binds to growth factors such as midkine (MK) and pleiotrophin (PTN) through its CS side chains (118). MK and PTN are present on radial glial fibers and serve as guidance cues for neuronal migration. The substrate for PTPζ has been identified as a G-protein coupled receptor kinase interactor-1 (GIT1, or Cat-1) (119). It is believed that binding of MK and PTN to PTPζ leads to receptor dimerization and inactivation of its phosphatase activity. This results in increased tyrosine phosphate level of GIT1 and leads to various cytoskeletal changes. This mechanism has been associated with growth promoting effects of CS, and highly sulfated CS such as CS-D and CS-E are involved in this pathway (120, 121). A splice variant of PTPζ called phosphacan promotes neurite outgrowth in rat mesencephalic and hippocampal neurons (57). It is thought that PTPζ dimerization leads to activation of various pathways such as mitogen-associated protein kinase (MAPK) and phosphatidylinositol 3-kinase (PI3-K) pathways (122, 123). In addition to its growth promoting effects, several studies claim that PTPζ is associated with CSPG-related inhibition in the CNS injury (124). Shen et al. show that after dorsal column injury, sensory axons grow deeper in PTPζ mutant mice than wild type mice. |