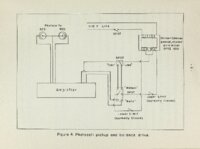
| OCR Text |
Show -40- as such, Using these constants, kjt and notations and k2'l equation (1) becomes, ltl CaCO:!::;;± CaO* (I']. [9] [1- superimposing specific reaction rate k2 This reaction involves a (2) CO2 + [Peo]· :2 measurable weight change as the gas leaves the solid, It is proposed that (CaO*) active lime changes to the cubic form of GaO according CaO* No weight changes portant as reaction reaction, Reaction step but the relative rates (3) is a diffusional process are or bulk CaO away from the surface, reversible the opposite reaction may also It is visualized, therefore, will form, part CaO under suitable CO2 q that The CaO which can the of the Since this process is occur, though much well-crystallized of the CaO will revert to the active form and conditions, im- whereby active CaO* leaves its position at the surface and become s part' crystalline (2) (3) noted in this noted below, to the by CaD are formed then, enter the carbonate lattice upon reaction with thus revert 'to active CaO* must be related to the concentration of the other reactants since it is also in the reaction zonae Its effective concentr-ation, in Iact, must be the centration of the uncovered potentially reactive CaC03' same as sites on the the con- |