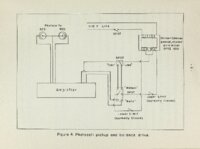
| OCR Text |
Show -39- l.q . Derivation of the theoretical rate equation: Thft classical stoichiometric expression of decomposition for the bulk reaction is, Caeoa· This reaction the calcium carbonate study, occur s as CaO well as at the surface take into account the area (1) COa + others, only (2,17) demonstrates that the actual and any rate study involved in the reactton, making the kinetic analysis it is considered and the final 3 is called here N, s.) zero It is to a an that CaO initially for med postulated that constant value as is original rhombohedral This intermediate CaO well-crystallized cubic CaO "active CaO" and is accordingly For the purpose of in some unstable structural state between that of the CaCO must (See given the designation, CaO*o' the concentration of the CaO* the reaction approaches incryases from steady state condtttons, The concentration of these groups is probably very small and is designated in the derivation below as, .6, the fraction of the total reactive surface covered by the active CaO*. Of the total surface involved in the reaction the .fraction occupied by the CaCOg is 1 ular groups which reactton, ·N.B.'NO .x a t e .the from ... 8, are It should be borne in mind that in position nference(is r a of ones those melee made that··this·CaO •. r this ·group. the\bulk CaO. s and··others still.attached'in related,to·the·····aetlvated (30).· s o a e way The· to a e the t t ve s u ... involved in thts The other inaccessible groups do not enter the reaction nGlienclature.used:by Eyring· r e to react are the only CllO· se a t e.v.Tn t sp t s e s b s t r a t e,' is OBly quite zone v r e a e t t on that different |