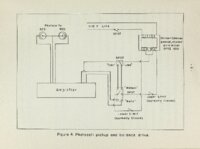
| OCR Text |
Show 25 - CaO formed; too slow CaO; too small a a or Phase equilibrium relationships have been 8120C(lO) to high have almost as which the CaCO s the the temperature size, shape, degree of calcin- deals with phase pressure-temper- pressure have been with more 14, 15) Methods involved sample' CaC0a- decomposition Equilibrium temperature values, CO2 9300C(11) 8900C (12, 13, exclusively on reported. atmosphere of around of on of the reactants and several for 1 being dependent of calcination Equilibria example, as of surface previous heating. CaO-C02 curves r ecarbonaticn may result in uncalcined carbonate independent Most of the literature ature permit Cunningham indicated that the line lump" force but is amount of 3. rate may constant linear rate which is gradient driving atton cooling temperature gradient in the center of the advances at a - a 'bomb' (a fine powder) reported as universally accepted used for type for low as values obtaining these results of pressure chamber in is caused to decompose, Pres. sures at equilibrium temperatures have been measured Smyth and Adams, (12) attached to the pressure chambers. quoted of the earlier and derived Tuttle, (16) 40 heating workers" used but not COOling by manometers a differential thermal curves and, according did not take into account the super most often analysis method to Harker and heating phenomena. Reaction Kinetics The kinetics of the reaction have received treatment in various British and French publtcattons, Whiting and Turner(17) found the rates |