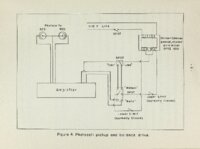
| OCR Text |
Show is the rate of R respectively; weight e temperature dependent and loss in nitrogen atmosphere; B experimentally deter mtned, is The mechanism of the decomposition is pictured stage phenomenon" Inttially the CO2 leaves the of CaO .apparently similar to the formation of thus acts , for this CaO from the active CaO(f rever shows the effect of water vapor on the Rate where At like B, is as a PH 2 0 catalyst _ =: BP value and AP H20 a + C02 explain ston, the the above of calcium carbonate C020 cases is expressed by, po cO2 1 is the pressure of water vapor. to assist in the removal of Significance Enthalpies as temperature dependent experimentally determined The Eyring absolute rate to The active CaO apparatus decomposition for these P C02 decomposition is the sible reaction. both in the presence and in the absence of 1+ two- a cell and the residue CaC03 Another set of experiments using the same The rate of being The second step of the reaction original CaCq,. bridge as metastable rhombohedral configuration assumes a well-crystallized as a is theory CO2 is The water vapor from the brought CaC03 in to play serves " in an attempt of the data in view of the above kinetic expre s- of activation and entropies of activation are estimated 'r - from the experimental data and their values add credence to the discusston. It is calculated that somewhat less than ular sites is one in 20,000 of the molec- responsible for the decomposition reaction and its observed rate. iii |