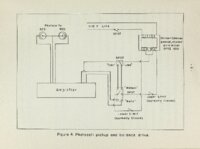
| OCR Text |
Show 32 - the original porous alundum tube with - a dense zircon tube of procelain very low gas per me abtltty, This tube were sealed with was an tightly fitted onto the gas connections and all A single atmosphere, exper imental isothermal furnace and the gas, of desired flow The balance .wi th temperature. The furnace operation, was raised change during that II - decrease area collected so was the to the desired set for automatic the tube surrounded the suspended weight) commenced, of undecomposed The a straight line of the 1 is a plot of a resulting time-weight in this typical during decomposition time-weight curves, a very calcium carbonate resulted the first 10 minutes of reaction Figure linearity brought were shape of the crystal sample only decomposttton, essentially during value, in the area of the initial therefore line rate, made in this way: Results Because of the tabular small run was sample suspended sample and collection of data (loss 20 The upper end of the asphaltic sealing compound, furnace tube remained open to the [oints curve was region and the slope used was rate curve, as It the of that weight loss was determined accounted for all of the non Table IT - 1 shows the data as • . The rates as determined above for each condition of temperature and carbon dioxide gas pressure Carbon dioxide gas pressures were were plotted as shown in Figure II - 2<) varied from 0 to about 645 mtlltrneter s |