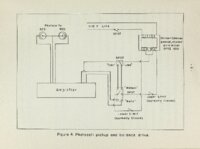
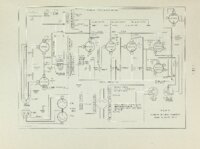
| OCR Text |
Show - to be influenced slow decomposition These authors moval of samples This latter runs, in to obtain tion of difficulty C0:t -free CaCOs was been given emphasis in obtaining determining importance Bretsznajder (2) .. , rates of decomposi involving periodic by Maskill measured, They and Turner(18) weight of an equation CO2 on of the first order; grains has three groups of investi to the endothermic reaction and also where early disintegra to from the surfaces of Inter-ior are re likewise unable were pieces of calcite due controlling step by components true rates of gave The rate found using results of work CO2 rate method mm as general, decreases maintained and the represented by as a Nucleation of the e, g mm 18, 19) Slf cooling due recombination of the was was in the furnace. Diffusion of released gator's, (2, air in 098 well weighing during decomposition was overcome good results with 3 precipitated a area as larger than Oq greatly increased handicapped by absorption tubes samples gave from the furnace for continuous flow of picked up attendant surface reproducibility while, mm were - Calcite grains with poor grain size below O. 098 tion, a size and the the conditions of heating, by in by grain 26 recognized as possible causing difficulty decomposttton, new CaO phase in the fine is also indicated as being of rate grained samples. (19) Zawadski and go further and show that the degree of to CaO, determines the rate of nuclei formation transformation, on the rever-sible r eaction, . Vallet and Richer(20) in recent work, hold that the rate determining |