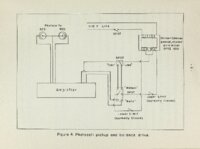
| OCR Text |
Show 31 - - Partial pressllres of carbon dioxide in the furnace atmosphere were obtained by mixing nitrogen and carbon dioxide in appropriate r atios, The nitrogen said to be "dry" but a was "dry" Drierite section was observed in the spectrographic examination, though counted for as CO 2 in an Or sat gas and 'as-received' gas did not reaction so it was was used to insure this condition" was small amount of water vapor red The carbon dioxide r-eceived, as at least analysts, original gas 99% - A very upon infra- of the gas was ac- of dried gas Interchange produce any noticeable changes in rates of concluded that the water vapor present in the gas was negltgtble, The temperature of the sample thermocouple the sample. in the with the junction Gas temperature pebble heater; temperature by an 2 equilibrium Checking on I CO2 winding a of similar kept was an inch from couple imbedded at the desired that was experiment This it equilibrium was was incor-rect, was CaCOs- in the literature. others(12)ere correct and into the furnace reaction zone, reported system proved pressures eration of all details of the reaching the measured by much lower than that a vacuum or's measurement of CO2 introduced one-eighth chr omel-ialume l a gave results which indicated that the ported by Smyth and Adams and the by electronic controller C1 was this in was measured than more The main furnace Early experiments CO not was data as re- that this auth- By careful consid- proved conclusively that being diluted in the system before corrected completely by substituting |