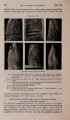
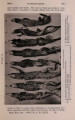
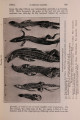
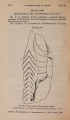
| OCR Text |
Show 88 DR. J. W. JENKINSON ON THE [Feb. 6, bath and left a brownish residue. On extraction with absolute alcohol the same yellow solution with a reddish tinge is obtained as before, giving the bands with exactly the same readings. " This alcohol solution was again evaporated down and the residue dissolved in ether, which dissolved a considerable portion, giving a yellowish solution with a reddish tint; this gave two bands :- (1) X 593 or 594-5 - 577, (2) \ 558 - 533-5. " This shifting is due to the influence of the solvent. "There also seems to be a slight shading between A 516 and 496? " This ether solution was evaporated down and the brown residue extracted with chloroform ; this formed a reddish solution, which contained oily-looking red drops floating undissolved on the surface. " The two bands now are :- (1) X 596 - 574, (2) A 558 - 533-5. 11 The first is not as dark as the second. " A faint greenish fluorescence seems to be present in all these solutions; it does not disappear on filtering. " The chloroform solution was next evaporated down and the residue dissolved in rectified spirit. " The addition of ammonia or of caustic soda to this solution causes precipitation, and, in the nitrate, diminishes the intensity of the colour of the solution and the shading of the bands, but does not appear to alter their position. On adding one drop of sulphuric acid the fluid becomes slightly turbid, and the two bands in the filtrate have disappeared to be replaced by two others, (1) before D and (2) in the green, which are difficult to measure, but whose position is approximately as follows:- (1) X 607 - 596, (2) X 568 - 547. " These recall at once the bands of acid hsematoporphyrin, the positions of which are :- (1) X 610-591, (2) X 585 - 567-5. " It is to be noted, however, that the addition of alkalis does not produce the usual four-banded spectrum of alkaline hsemato-porphyrin, but Garrod and others have described in urine a two-banded neutral hamiatoporphyrin spectrum. " Fearing that possibly a four-banded alkaline hamiatoporphyrin spectrum might have been present but have been passed over owing to the weakness of the solution, a layer of fluid (absolute |