| Title |
Congenital Mydriasis Associated With Megacystis Microcolon Intestinal Hypoperistalsis Syndrome |
| Creator |
McClelland, Collin; Walsh, Ryan D; Chikwava, Kudakwashe R; Johnson, Mark P; Mattei, Peter; Liu, Grant T |
| Affiliation |
Department of Ophthalmology and Visual Sciences (CM), Washington University, St Louis, Missouri; Departments of Ophthalmology and Neurology (RDW), The Medical College of Wisconsin, Milwaukee, Wisconsin; The Neuro-Ophthalmology Service (GTL); Department of Pathology (KRC); Center for Fetal Diagnosis and Treatment (MPJ); Division of General, Thoracic and Fetal Surgery in the Department of Surgery (PM), The Children's Hospital of Philadelphia and The Perelman School of Medicine, University of Pennsylvania, Philadelphia, Pennsylvania |
| Abstract |
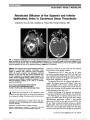
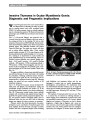
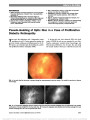
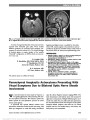
We report a case of congenital mydriasis in a neonate with megacystis microcolon intestinal hypoperistalsis syndrome (MMIHS). Pilocarpine testing and gastrointestinal pathology in our patient suggest that the mydriasis is due to an underlying smooth muscle myopathy of the iris sphincter muscle. These findings may have important implications regarding the pathogenesis of MMIHS. |
| Subject |
Abnormalities, Multiple; Colon; Eye Diseases, Hereditary; Female; Humans; Intestinal Pseudo-Obstruction; Muscle, Smooth; Mydriasis; Urinary Bladder; Young Adult |
| Format |
application/pdf |
| Publication Type |
Journal Article |
| Collection |
Neuro-Ophthalmology Virtual Education Library: Journal of Neuro-Ophthalmology Archives: https://novel.utah.edu/jno/ |
| Publisher |
Lippincott, Williams & Wilkins |
| Holding Institution |
Spencer S. Eccles Health Sciences Library, University of Utah |
| Rights Management |
© North American Neuro-Ophthalmology Society |
| Setname |
ehsl_novel_jno |
| ID |
227486 |
| Reference URL |
https://collections.lib.utah.edu/ark:/87278/s6gr03wb/227486 |