| OCR Text |
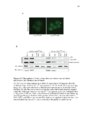
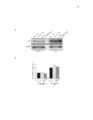
Show 28 Molecular Cell Mitochondrial Stress and Protein Degradation a C-terminal VIM sequence and the interaction with Npl4 is mediated by Cdc48. Specifw:; loss ofCdc48and Npl4 binding inthis mutant enabled us to test whether the genetic function of Vms1 requires interaction with Cdc48 and Npl4. We assayed the ability of wHd-type and VIM mutant VMS1, expressed from the native promoter, to rescue the rapamycin hypersensitivity of the vms16. mutant strain. ~ shown in Figure 50, wild-type Vms1 rescued fully, but the VIM mutant Vms1 had no effect on growth despite being expressed at levels equivalent to wild-type Vms 1 (see Figure 5C). These data suggest that the function of Vms1 required for rapamycin resistance, which we hypothesize relates to protection of mitochondrial functio n from oxidative danage (see Figure 2), is completely dependent upon interaction with Cdc48. To identify the protein interactions of mammalian Vms1, we conducted a tandem-affinity purification of a Flag/HA-tagged mouse Vms1 expressed in mouseC2C12cells. The final elution had essential y only two bands (Figure 5E), Vms1-Flag/HA and VCP, the mammalian ortholog of Cdc48. This interaction was confirmed by coimmunoprecipitation experiments. WhHe wWdtype Vms1 precipitated endogenous VCR (Figure 5F, lane 6), this interaction was completely lost in a mutant wherein three highly conserved VIM residues (as indicated in Figure SA) had been mutated to alanine (Figure 5F, lane 9). This experiment was performed quantitatively to enable determination of the amount of VCP that associates with Vms1. While greater than 90% of the tagged Vms1 was precipitated, we observed essentially no depletion of the total VCP (Figure 5F, compare lanes 4-6), suggesting that the Vms1-associated VCP accounts for less than 5% of the total cellular VCR. The yeast Vms1 also appears to interact with a similarly small fraction of the total cellular Cdc48 (data not shown). This is consistent with the observation that Cdc48 has a much higher abundMce than Vms1 in normal yeast cells (Ghaemmaghami et aI., 2(03). Vms1-Dependent Mitochondrial Translocation of Cdc48 Based on the stable and constitutive interaction between Vms1 and both Cdc48 Md Npl4, we hypothesized that Vms1 might mediate the recruitment of a subset of Cdc48 and Npl4 to mitochondria. Under normal conditions, Cdc48-GFP localized throughout the cytoplasm, nucleus, and ER (Figure 6A). Upon hydrogen peroxide treatment, however, a fractio n of Cdc48GFP translocated to mitochondria in wild-type cells (Figure 6A). The stress-induced mitochondrial translocation of Cdc48 was nearly absent in a vms16. mutant strain, but was restored by a plasmid-borne copy of VMS1 (Figure 6A). Cdc48-GFP also exhibited localization to nonmitochondrial punctae upon hydrogen peroxide treatment that was independent of Vms1. The ratio of Cdc48-GFP that coIocalized with mito-RFP versus total cellular Cdc48-GFP was blindly quantitated in these three strains in the presence and absence of hydrogen peroxide. This quMtitative analysis showed that loss of VMS1 largelyabrogated the peroxide-induced mitochondrial localization of Cdc48GFP (Figure 6B). There appears to be, however, a small fractio n of Cdc48 that shows stress-responsive mitochondrial coIocalization that is independent of Vms1. Our imaging Md quantification are currently unable to distinguish whether t his is due to an alternative mitochondrial-targeting system or to Cdc48 Ioca~za- tion to sites neart1y' mitochondria. A fractio n of Npl4also exhibits stress-responsive translocation to mitochondria that is dependent on Vms1 (Figure 56). If Vms1 recruits Cdc48 and Npl4 to increase their local concentration at mitochondria in response to stress, overexpression of Npl4 and/or Cdc48 might partially complement the vms16. mutant phenotype by nonspeciflCSlly increasing their local concentration at mitochondria. As expected, both Cdc48 and Npl4 overexpression suppressed the rapamyci n hypersensitivity of the vms16. mutant strain (Figure 6C). Consistent with the observation that Ufd1 does not associate with Vms1, overexpression of Ufd1 had no effect on the vms 16. mutant phenotype (Figure 6C). Together, these data support a role for Vms1 in recruiting Cdc48 and Npl4 to mitochondria in responseto stress, and this activity is a primary function of Vms1. VMS1 Is Requiredfor Mitochondrial Protein Degradation As part of the ERAO machinery, Cdc48 and Npl4 promotethe retrotranslocation of lumenal ER or ER membrane proteins to the cytosol where they are degraded by the proteasome in a ubiquitin-dependent manner (Raasi and WoIf,2007; Vembar and Brodsky, 2(08). Under the hypothesis that the Vms1-Cdc48-Npl4 complex functions similarly at mitochondria, we measured the steady-state levels of Fzo1. Fz01 is a mitochondrial outer membrane protein that is the best-characterized mitochondrial ta-get of ubiquitin/proteasome system-dependent degradation in yeast (Cohen et aI., 2008; Escobar-Henriques et aI., 2(06). While the levels of Fz01 are identical in wild-type and the vms16. mutant in log phase (data not shown), the vms 16. mutant exhibited elevated Fz01 levels at day 2.5 of culture (Figure 7~. The levels of Fz01 were similar to that found in a mutant lacking Mdm30, the principal ubiquitin E31igase involved in Fz01 degradation (Cohen et aI., 2(08). The mdm306. vms 16. double mutant showed M additive effect (Figure 7A). If the Vms1-Gdc48-NpI4 complex is req uired for normal Fz01 degradation, mutants of Cdc48 and Npl4 should also show elevated Fz01 staooy-state levels. The S565G mutant of Cdc48, which exhibits impaired Vms1 binding, had elevated Fz01 levels simila- to the vms16. mutant (Figure 7B). The Fz01 levels of the vms 16. cdc48-5565G double mutant are similar to the two single mutants, consistent with the proteins acting within the same pathway to promote Fz01 degradation. Fz01 also accumulated in the temperature-sensitive np14- 1 mutant at the permissive temperature (Figure 5 7~, where there is no growth defect on giucose-containing medium (see Figure 57B). Consistent with a role for Npl4 in supporting mitochondrial function, the np14- 1 mutant grew poorly on glycerol medium, but not glucose medium (Figure 57B). To directly address the stability of Fz01, we examined the degradation kinetics of an Fzo1-HA fusion protein fonowing cydoheximide addition to prevent new protein synthesis. Degradation of Fz01-HA was reproducibly delayed in the vms16. mutant relative to wHd-type cells (Figure 7C - quantitation in Figure 57C). Under identical experimental conditions, the ERAD substrate CP'(· was degraded with similar kinetics in the wHd-type and vms16. mutant strains (Figure 70). In fact, the degradation of CRY· in the vms 16. mutant was slightly enhanced relative to wild-type (Figure 70), w hile CP'(· was stabiWzed in Molecular Ceil 40, 465-460, November 12, 2010 C2010 Elsevier Inc. 471 |