| OCR Text |
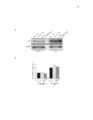
Show 26 Molecular Cell Mitochondrial Stress and Protein Degradation colony formation on glycerol was nearly absent (Figure2E). After 8.5 days of culture, the ~s1 tJ. mutMt also showed greatly reduced ability to form colonies on glucose medium (Figure 2E). This is likely due to cell death, as we obtained similar results using exclusion of trypan blue as a measure of viability (data not shown). Two independent vms 1tJ. isolates in adifferentstrain background (BY4741) exhibited an even more rapid loss of viability than was observed in the W303 ba:::kground (Figure 2F). Oxidative stress has been proposed to be the key mediator in causing cell death in static culture (Fabrizio et aI., 2(01 ), and the vms 1tJ. mutant showed a significant increase in the oxidation of DHE to ethidium (Figure 2G and Figure S2A). In addition to measuring mitochondrial function and glycerol growth in static culture, we also examined the effect of oxidative stress. As with growth to day 1.5, combination with the sod2tJ. mutMt caused a significant loss of oxygen consumption and aconitase activity in the vms1tJ. mutant (Figures S2B and S2C). Similarly, while the vms1tJ. mutMt exhibited normal glycerol growth in the absence of additional stressors, combination with a deletion of SOD2 caused a complete loss of growth on glycerol medium (Figure 2H). Combined, these data show that the vms1tJ. mutant has severely compromised mitochondrial activity under the conditions that cause Vms1 mitochondrial translocation. VMS1 Localization and Function in C. e/egans To determine whether VMS1 orthologs from other eukayotes function similarly, we examined vms- 1 in C. elegans, which contains a single VMS1 ortholog encoded by K06H7.3. Either of two nonoverlapping RNAi constructs targeting vms- 1 caused a marked reduction in viabi~ty in response to hydrogen peroxide (Figure 3A). The surviving vms- 1-depleted individuals were dramatically more lethargic than controls. This was confirmed in a mutMt line carrying a deletion predicted to remove the majority of the VM5-1 protein (data not shown). Under standard growth conditions, vms- 1 mutants and RNAi-treated animals had normal morphology and wWd-type growth and development. P(:tivation of numerous stress-response genes, including the mitochondrial superoxide dismutase genes sod-2 and sod-3, is dependent on the functio n of the insulin-regulated FOXO transcription factor DAF-16 (Murphy et aI., 2003; Oh et aI., 2(06). As expected, daf- 16 mutant animals were also hypersensitive to hydrogen peroxide treatment (Figure 3A). Treatment of daf16 mutants with vms- 1 RNAi caused further hypersensitivity of similar relative magnitude to that observed in wild-type vms- 1 RNAi-treated animals (Figure 3A). Knockdown of vms- 1 also caused a significant decrease in life span of wild-type animals and a further decrease in the already shortened life span of daf- 16 mutants (Figure 3 B). These findings suggest that vms- 1 functions in parallel with insulin signaling to regulatestress resistance and life span. To determ ina the tissue and subcellular expression patterns of C. e/egans VMS-1, we generated transgenic lines in w hich fullength VMS-1 fused to a GFP reporter was expressed from the native vms- 1 promoter. This reporter was expressed broady during embryonic development. In larval stages and in adults, expression was noted in intestinal cens, specific neurons in the head Md the taW, and in the ventral nerve cord (Figure 3C and Figure 53). In untreated Mimals, VM5-1::GFP loca~zed to the cytoplasm in intestinal cells and neurons. In head amphid neurons, VM5-1::GFP was uniformly detected in the dendritic processes, where it was specifically excluded from mitochondria, as determined by lack of coIocalization with DICl::mCheny (Kass et aI., 2(01) (Figure 3C). Exposure of animals to hydrogen peroxide, however, caused coIocalization of VMS1::GFP with DIC-1 ::mCheny (Figure 3C), an identical pattern to that seen for other mitochondrial proteins (Hu and Barr, 2(05). Together, these findings indicate that, as in yeast, C. elegans vms- 1 functio n is dispensable for viability and growth but is required for protection against oxidative stress and for wildtype life span. Vms1 Constitutively Interacts with Cdc48 and Npl4 WhiletheVms1 protein expressed inE. coli migrated asa monomer in gel filtration chromatography, endogenous Vms1 from crude yeast Iysates migrated in a large >500 kDa complex (data not shown). To identify subunits of the putative Vms1 complex, we purified a functional Vms1-TAP fusio n and identified associated proteins by mass spectrometry. Cdc48, a hexameric AM-ATPase with a well-studied role in protein degradation (Jentsch and Rumpf, 2(07), copurified almost stoichiometrically with Vms1-TAP (Figure 4A). The Vms1-Gdc48 interaction was confirmed by coimmunoprecipitation ofepitope-tagged versions ofVms1 and Cdc48expressed under their endogenous promoters. Immunoprecipitation of Vms1-HA pulled down Cdc48-myc (Figure 4B). This interaction was also observed following 2 hr treatment with rapamycin (Figure S4A), a condition that causes Vms1 mitochondrial translocation. The S565G mutant of Cdc48 was previously reported to cause increased sensitivity to oxidative stress, reduced respiratory activity, and increased cell death (Braun et aI., 2006; Madeo et aI., 1997). This mutant was expressed at wWd-type levels, but interacted much more weakly with Vms1 than did wild-type Cdc48 (Figure 4B). Thus, Vms1 exists in a stable complex with Cdc48, and this interaction is disrupted by a Cdc48 mutation associated with increased oxidative stress sensitivity and cel death. Cell death in Cdc48-8565G mutant strains has been previously shown t1t increased annexin V and propidium iodide (PQ staining (M adeo et al., 1997). Uke Cdc48S565G mutant strains, the vms1tJ. mutMt strain had a significantly increased fractio n of Armexin V and PI-positive cens (Figures S4B and S4C). (0) WT Ilfld """$1 01. $lmin$ in the BY4741 baclqWldw,,",e {T0Wfl in synthet ic ~te g lucOSEl (SO) media for 4 daysllfld stained with ditJy(lroethidium. Ethidium ftuOffl!lCetl(:e wasdE!termined by FACS Ilflaly$i$ for each strain. For$lch $lrain , ttr""" independent c<Jtures _e te$led Ilfld mellfl ± SO i$$hCWn. (11 WT, """$1 01., $Cd201., 1lfld vl7I$l 01. $Cd201. $lmin$ _e trllfl$lormed wittl ef1l)lyVEl(:tor (ev~ a pIa$"11 id containing the SOD2 gene (pSOO2) or a plasmid CO'ltaining tile IIMSI gene ~VMSI ~ Each strain wasQrcwn to sahraticl'l in SO-IJra media. 5eria I5-folddilulion$ Of each c<J!"..e _e tIlen spotted on an SOlycerol-Um plate and grown at 30'C. Molecular Ceil 40, 465-460, November 12, 2010 C2010 Elsevier Inc. 469 |