| OCR Text |
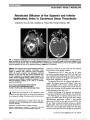
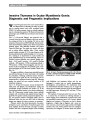
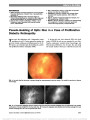
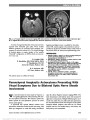
Show Nonarteritic Anterior Ischemic Optic Neuropathy and Obstructive Sleep Apnea Gorkem Bilgin, MD, FEBO, Yaran Koban, MD, Anthony C. Arnold, MD Background: To evaluate the obstructive sleep apnea syndrome (OSAS) in patients with nonarteritic anterior ischemic optic neuropathy (NAION). Methods: We recruited 27 patients with NAION and 27 age-matched and sex-matched controls who also were similar for systemic risk factors such as diabetes mellitus, hyper-tension, and hypercholesterolemia. All patients and controls underwent overnight polysomnography for the diagnosis of OSAS and calculation of apnea-hypopnea index (AHI). Results: Patients and controls were statistically similar in terms of age, sex, gender, smoking, systemic risk factors, neck circumference, and body mass index. The subjects with AHI $ 20 were accepted as OSAS. Fifteen of 27 patients (55.6%) with NAION and 6 of 27 controls (22.2%) had OSAS (P , 0.05). Conclusion: The prevalence of OSAS was higher in patients with NAION, and the difference between patient and control groups was statistically significant (P , 0.05). This result supports prior series suggesting the association between NAION and OSAS. Journal of Neuro-Ophthalmology 2013;33:232-234 doi: 10.1097/WNO.0b013e31828eecbd © 2013 by North American Neuro-Ophthalmology Society Nonarteritic anterior ischemic optic neuropathy (NAION) is a common cause of visual loss from optic nerve dysfunction. It is characterized by sudden or rapidly progressive, monocular or binocular visual decline, initially accompanied by segmental or diffuse optic disc edema, and later optic atrophy and retinal arteriolar narrowing (1,2). NAION is thought to result from circulatory insufficiency within the optic nerve head, but the specific mechanism and location of the vasculopathy remain unknown (3). Obstructive sleep apnea syndrome (OSAS) is a common yet underdiagnosed condition that may be associated with significant morbidity if left untreated. It is characterized by recurrent interruption of normal breathing during sleep, owing to upper airway obstruction (apneic spells) (4). The apneic spells can cause a decrease in the arterial oxygen saturation and an increase in the carbon dioxide saturation during sleep. OSAS has been associated with an increased risk of cardiovascular disease, hypertension (HT), and stroke (5-7). We evaluated the possible association between OSAS and NAION. METHODS This prospective study involved 27 patients withNAIONwho were evaluated within 1month of symptomonset. The control group consisted of 27 age- and sex-matched subjects with similar systemic risk factors for disorders such as diabetes mellitus, HT, and hypercholesterolemia. This study followed the tenets of the Declaration ofHelsinki, and informed consent was obtained from all subjects. The study was approved by the Erzurum Regional Education and Research Hospital Ethical Committee and Review Board. The diagnosis ofNAIONwas made based on the following criteria: 1. Sudden onset of painless visual acuity and/or visual field loss 2. Diffuse or sectoral optic disc edema 3. Presence of relative afferent pupillary defect 4. Lack of clinical findings suggesting another disorder. Polysomnography Participants underwent overnight polysomnography (PSG) recordings in 2 sleep laboratories. Sleep was continuously recorded on a computerized system (Grass Technologies, West Warwick, RI) scored in 30-second epochs according to the American Academy of Sleep Medicine standardized criteria (8). PSG measurements included electroencephalograms Jules Stein Eye Institute (GB, ACA), Neuro-Ophthalmology Division, University of California, Los Angeles, Los Angeles, California; and Ophthalmology Division (YK), Bayburt State Hospital, Bayburt, Turkey. The authors report no conflicts of interest. Address correspondence to Gorkem Bilgin, MD, FEBO, Jules Stein Eye Institute Room 2-247; 100 Stein Plaza, Los Angeles, CA 90095- 7005; E-mail: gorkemgencebilgin@gmail.com 232 Bilgin et al: J Neuro-Ophthalmol 2013; 33: 232-234 Original Contribution Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. (EEG; C3-A2, C4-A1, O1-A2, O2-A1, F3-M2, F4-M1), right and left electrooculograms, mental-submental electro-myogram (EMG), electrocardiogram, leg EMG, thoracic and abdominal wall motion (respiratory inductance plethysmogra-phy), pulse oximetry, combined nasal/oral thermistor, and a body position detector. Apnea during sleep was defined as cessation of airflow (90% fall in the amplitude of airflow signal compared to the baseline airflow) lasting at least 10 seconds. Hypopnea was defined as a 50% or greater fall in airflow lasting 10 or more seconds associated with a 3% or greater fall in oxygen saturation from baseline. The apnea-hypopnea index (AHI) was calculated using the total number of respira-tory events (apneas and hypopneas) per hour sleep (9). Sub-jects with an AHI $20 were regarded as having OSAS. Statistical Analysis Means were compared by Student t test or Mann‐Whitney U test. Prevalence of OSAS in patients with NAION was compared with the matched controls using Fisher exact test. Comparison of the clinical and PSG characteristics of the patients with NAION and matched controls was performed using the unpaired t test or Fisher exact test. SSPS version 17.0 for Windows (SSPS, Inc, Chicago, IL) was used for statistical analysis. Significance was accepted for P , 0.05. RESULTS Mean age of the patients with NAION and matched controls were 64.9 ± 7.86 years and 63.7 ± 5.24 years, respectively. There were no statistically significant differences with regard to age, gender, systemic risk factors, smoking, neck circum-ference, and body mass index between the 2 groups (P . 0.05). These data are summarized in Table 1. Fifteen of 27 patients (55.6%) with NAION and 6 of 27 subjects (22.2%) in the control group met the criteria for OSAS (AHI $ 20); the difference was statistically significant (P , 0.05, Fisher exact test). Relative risk for sleep apnea in NAION patients was 2.5 compared to the subjects in the control group. DISCUSSION We found an increased prevalence of OSAS in patients with NAION. Several previous studies suggest the association. Hayreh (10) reported several patients with anterior ischemic optic neuropathy having a history of OSAS. Li et al (11) found that the prevalence of OSAS was 30% in 73 newly diagnosed patients with NAION and 17.8% in 73 age- and sex-matched controls. However, they used the survey of Sleep Apnea Scale of Disorders Questionnaire for the diag-nosis of OSAS instead of PSG. Mojon et al (12) recruited 17 patients with NAION, compared them with 17 age- and sex-matched controls, and performed overnight PSG on each patient. Twelve of 17 NAION patients (71%) and 3 of 18 controls (18%) had sleep apnea (P , 0.001). In this study, they used respiratory disturbance index (RDI) .10 in establishing the diagnosis of OSAS. Arda et al (13) studied 20 patients with newly diagnosed NAION and 20 age- and sex-matched subjects with similar systemic risk factors for NAION as control group. All patients and controls underwent overnight PSG. Cases with an AHI .5 were accepted as having OSAS. The prevalence of OSAS was found to be 85% and 65% in the patient and control groups, respectively. There was no statistically significant dif-ference between the 2 groups. The diagnosis of OSAS is definitively established by nocturnal PSG (14). The definition of obstructive events is reported as AHI or RDI in PSG reports. AHI reflects the number of apneas and hypopneic episodes per hour of sleep (9). On the other hand, in addition to apneas and hypopneas, RDI includes breathing disturbances that are not included in the definition of apnea and hypopnea, that are abnormal without clear impact on oxygen saturation, or clear, visually recognizable EEG arousal (14). In other words, RDI can be higher or equal to AHI. OSAS severity is defined as mild for RDI $5 and ,15, moderate for RDI $15 and #30, and severe for RDI .30 per hour (9). In our study, we considered the subjects with an AHI $ 20 as having OSAS. Mojon et al (12) and Arda et al (13) accepted RDI .10 and AHI .5, respectively, as the threshold to diagnose OSAS, thus including subjects with mild or mild-moderate OSAS. Palombi et al (15) reported that 89% of 27 patients with newly diagnosed NAION had an AHI .15, concluding that NAION was nearly always associated with sleep apnea. We believe that by using more stringent criteria of AHI rather than RDI and a cutoff point of 20, we were able to reduce false-positive findings and provide a more accurate assessment of prevalence. TABLE 1. Clinical characteristics of patients with NAION and control group Patients With NAION (n = 27) Controls (n = 27) Age, mean ± SD (y) 64.9 ± 7.86 63.7 ± 5.24 Sex, n (%) Male 15 (55.6) 15 (55.6) Female 12 (44.4) 12 (44.4) Smoking 15 (55.6) 14 (51.9) Body-mass index, mean ± SD (kg/m2) 28.9 ± 2.3 27.5 ± 3.6 Neck circumference, mean ± SD (cm) 35.97 ± 4.61 37.43 ± 3.78 Systemic disease, n (%) Hypertension 15 (55.6) 14 (51.9) Diabetes mellitus 7 (26) 6 (22.2) Hypercholesterolemia 12 (44.4) 10 (37) Coronary artery disease 5 (19) 3 (11.1) NAION, nonarteritic anterior ischemic optic neuropathy. Bilgin et al: J Neuro-Ophthalmol 2013; 33: 232-234 233 Original Contribution Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. In conclusion, OSAS should be considered as a signifi-cant risk factor for NAION. Because there is no proven treatment for NAION, control of risk factors may be important in preventing involvement of the second eye. At this point, PSG should be considered in every patient with newly diagnosed NAION. REFERENCES 1. Rizzo JF, Lessell S. Optic neuritis and ischemic optic neuropathy: overlapping clinical profiles. Arch Ophthalmol. 1991;109:1668-1672. 2. Boghen DR, Glaser JS. Ischemic optic neuropathy: the clinical profile and natural history. Brain. 1975;98:689-708. 3. Arnold AC. Pathogenesis of nonarteritic anterior ischemic optic neuropathy. J Neuroophthalmol. 2003;23:157-163. 4. Guilleminault C, van den Hoed J, Mitler MM. Clinical overview of the sleep apnea syndrome. In: Guillminault C, Dement WC, eds. Sleep Apnea Syndrome. The Kroc Foundation Series, Vol. 2. New York, NY: AR Liss, 1978:1-12. 5. Lattimore JD, Celermajer DS, Wilcox I. Obstructive sleep apnea and cardiovascular disease. J Am Coll Cardiol. 2003;41:1429-1437. 6. Mohsenin V. Sleep-related breathing disorders and risk of stroke. Stroke. 2001;32:1271-1278. 7. Pepperell JC, Davies RJ, Stradling JR. Systemic hypertension and obstructive sleep apnea. Sleep Rev Med. 2002;6:157-173. 8. Iber C, Ancoli-Israel S, Chesson AL, Quan SF; for the American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications. Westchester, IL: American Academy of Sleep Medicine, 2007. 9. Epstein LJ, Kristo D, Strollo PJ Jr, Friedman N, Malhotra A, Patil SP, Ramar K, Rogers R, Schwab RJ, Weaver EM, Weinstein MD. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5:263-276. 10. Hayreh SS. Acute ischemic disorders of the optic nerve. Pathogenesis, clinical manifestations and management. Ophthalmol Clin North Am. 1996;9:407-442. 11. Li J, McGwin G Jr, Vaphiades MS, Owsley C. Non-arteritic anterior ischaemic optic neuropathy and presumed sleep apnea syndrome screened by the Sleep Apnea scale of the Sleep Disorders Questionnaire (SA-SDQ). Br J Ophthalmol. 2007;91:1524-1527. 12. Mojon DS, Hedges TR III, Ehrenberg B, Karam EZ, Goldblum D, Abou-Chebl A, Gugger M, Mathis J. Association between sleep apnea syndrome and nonarteritic anterior ischemic optic neuropathy. Arch Ophthalmol. 2002;120:601-605. 13. Arda H, Birer S, Aksu M, Ismailogullari S, Karakucuk S, Mirza E, Gumus K, Oner A. Obstructive sleep apnea prevalence in non-arteritic anterior ischaemic optic neuropathy. Br J Ophthalmol. 2013;97:206-209. 14. Benbir AG, Guilleminault C. Obstructive and nonobstructive sleep apnea: the neurological perspective. In: Culebras A, ed. Sleep Disorders and Neurological Diseases, 2nd edition. New York, NY: Informa Healthcare USA Inc, 2007:277-299. 15. Palombi K, Renard E, Levy P, Chiquet C, Deschaux Ch, Romanet JP, Pepin JL. Non-arteritic anterior ischaemic optic neuropathy is nearly systematically associated with obstructive sleep apnoea. Br J Ophthalmol. 2006;90:879-882. 234 Bilgin et al: J Neuro-Ophthalmol 2013; 33: 232-234 Original Contribution Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. |