| OCR Text |
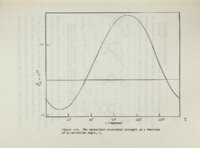
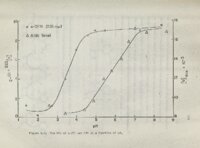
Show 134 N3 and could associate directly with this atom a solvent are bridge. In additon the or indirectly through hydrogens attached to C'2 and C'3 capable of forming weak hydrogen bonds according to a recent study on similar conformation at systems?O The C2 hydrogen qould help stabilize cn = +300 being coplanar with Very weak hydrogen bonds of a kilocalorie nificant Such II N3 at this angle, or even hydrogen bonds should become more protic orgnic solventsi as a less could be in stable sig- weakly the dielectric constant of.the media is reduced, The hydrophobic forces may, however, be the predominant stabilizing Organic solvents feature. cause extensive acids. It is as dioxane or such as formaldehyde have been found to disruption of the hydrophobic forces in helical nucleic probi!lble that solvents with 1T-.electron systems such acetonitrile would reduce the interaction between the This is 1T-system of the sugar group and the base chromophore. possible explanation of why WB in water and methanol are a approximately equal ("'55)., ,while B in dioxane and acetonitrile have values ar-ound 18. In Significant has a 8Ql it is observed that solvent effects Figure foJ'l III which has the is that there ae much mOJ'le C.S thioehter group than for II which hydroxyl function at this position. pJ'letation are The most reasonable inter- important conformational equilibriums involving the tl)ioehter group as well as those involving the sugar's ! ( ;' |