| OCR Text |
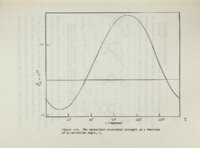
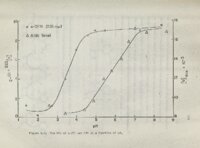
Show 94 Yo is also apparent from It Figure 506 that protonation of site induce the largest rotation of Yo N3 will o In 6-substtuted (pKa 's 2-4)0 In purines Figure 302 it is know that site the ORD of curves Nl is protonated 71 6=chloropurine and 6-amnopurine run with pH values two unis above"and below the proper show very little pK values 5'05 and 5 Q 6 t a sUght pH effect., although, increase on the bases of might have been expect ed \ , Figure Similarly 1 quanosine72.which has a,pKa involving N7,and its 8=substituted de\ rivatives ·show no cyclopurine nucleosides (Figures 401=5) effects on the Cotton effects of protonation has very little w effect showed no w* origino on w on the significant pH It appears that w* transitions possibly protonation does not significantly increase the spectroscopic because moment Moreover the data significant pH effectso of the The aza-nitrogeno pH effects when they do occur can best be rationalized in s: terms of C_otton effects arising from n w" transition that are blue I .'. shifted upon pr-o'tonat Ion , Normally Tr -+ 'It" transitions dominate ultraviolet range and it is reasonable to assume ';': Cotton effects lie blue shifts upon the case submerged under the Tr n Tr* Cottpn effects and their protonation often go undetectedo is revealed of adenosine Tr that the small the That this is likely by a comparison of the absorption and ORD s.pectra I and 3-deazadenosine 110 |