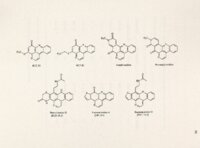
| OCR Text |
Show 60 radiolabel enough DNA for 10 subsequent cleavage reactions, 10 JlI pUC 19 DNA (1.5 ug) was aliquoted into a 0.5 ml microfuge tube and mixed with 10 JlI H20 and 5 JlI 1 M NaOR To this, 2.5 JlI of primer (5 pmol) was added with 10 min incubation on ice. Then 1 M HCI (5 JlI) was added along with 5 ul labeling reaction buffer (400 mM Tris HCI, pH 7.5, 100 mM MgCl2 and 250 mM NaCI), the mixture incubated again at 37°C for 10 min, and placed on ice. DTT (2.5 JlI of a 0.1 M stock) was added next with 5 JlI of labeling mix (1.5 JlM dCTP, dGTP and dTTP), 1.25 JlI [32p]dATP (10 mCilml) and 2.5 JlI diluted Sequenase 2.0 (IJlI of Sequenase, 13 U, plus 1 JlI of pyrophosphatase, 5 U and 6 JlI of glycerol enzyme dilution buffer, 20 mM Tris-HCI, pH 7.5, 2 mM DTT, EDTA and 50% glycerol). This mixture was incubated for 5 min at room temperature. The reaction was chased with 25 JlI chase mix (1.7 mM of each deoxynuc1eotide triphosphate in H20) and 7.5 JlI of additional diluted Sequenase 2.0, for 6 The reaction was 0.1 mM stopped by the addition of 40 JlI each min at 37°C. of phenol and then centrifuged for 15 chloroform:isoamyl alcohol (24:1) with mixing. The reaction was to min. at 15K x g. Then 65 JlI of the aqueous phase (80% of the volume) was transferred a second microfuge tube to which 130 JlI of ice-cold ethanol, 2.5 JlI of 5 M NaCI and 1 JlI on 1 M MgCl2 were added and mixed. Radiolabeled DNA was precipitated by freezing solid CO2 for 20 min and centrifugation at 40°C, 15K x g for 20 min. Ethanol was aspirated, 130 JlI of ice-cold 70% ethanol were added with mixing, freezing on solid CO2 and for 5 min and centrifugation at 40°C, 15K x g for 5 min. Ethanol was again aspirated the labeled DNA was allowed to dry in air. |