| OCR Text |
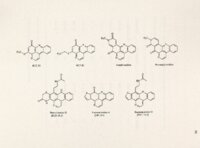
Show 56 Protective effects certain against ascididemin-induced DNA cleavage were antioxidants, antioxidant enzymes and radical scavengers. afforded by Lack of additional stimulation of DNA cleavage by metal ions and only slight protection with a chelator (at very high concentrations) excluded a Fenton-type reaction as the primary mechanism of DNA cleavage. Extensive protection against DNA cleavage by the antioxidant enzyme catalase, responsible for the 'detoxification of H202 to H20, suggested H202 as the possible reactive species and further supported the role of ROS in ascididemin DNA cleavage. Because protection was varied among antioxidants and scavenging chemicals, pHs of the various reactions were measured to ensure that pH had no effect on protection competency. Because the reactions covered a range of pHs from 3.56-9.27, cleavage reactions run in a series of Tris-HCl buffers of pH range 3-10 confirmed there DNA was cleavage by the test compounds or protection essentially no effect of pH on capabilities oyer this range. In addition to catalase, slight to good protection was also evidenced by the in vitro addition of antioxidants glutathione, BHA and NAC and the radical scavenger benzoic acid. Thus, these results implicate an oxidative mechanism in ascididemin DNA cleavage. The production of ROS likely occurs through reduction of the imino quinone structure to a semiquinone which could facilitate the production of DNA damaging free radicals. Finally, DNA cleavage in vitro by ascididemin (BC-I-51), BC-I09-1 and BCMH-l48 required oxygen. oxygen. Similarly, redox cycling and ROS generation require a source of Under anaerobic conditions, ascididemin, BC-I09-1 and BCMH-1-48 lost their ability to cleave DNA. This was true even in the presence of DTT and incubation |