| OCR Text |
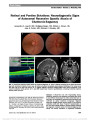
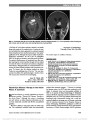
Show Retinal and Pontine Striations: Neurodiagnostic Signs of Autosomal Recessive Spastic Ataxia of Charlevoix-Saguenay Jacqueline A. Leavitt, MD, Wolfgang Singer, MD, William L. Brown, OD, Jose S. Pulido, MD, Michael C. Brodsky, MD Abstract: A 39-year-old man with long-standing ataxia, spasticity, dysarthria, and peripheral neuropathy was found to have diffuse thickening of the retinal nerve fiber layer in both eyes, as manifested by prominent retinal striations and confirmed by optical coherence tomography. Magnetic resonance imaging showed severe atrophy of the superior cerebellar vermis with linear "footprint" hypointensities in the pons with irregular striations. Genetic testing confirmed the diagnosis of spastic ataxia of Charlevoix-Saguenay (AR-SACS). The clinical evaluation of progressive cerebellar FIG. 1. Autosomal recessive spastic ataxia of Charlevoix-Saguenay. A. There is diffuse thickening of the retinal nerve fiber layer with prominent striations in each eye. B. Optical coherence tomography demonstrates thickening of the retinal nerve fiber layer next to the optic disc (arrow). C. T1 sagittal magnetic resonance imaging (MRI) shows severe hypoplasia of the superior cerebellar vermis (arrow). D. T1 axial MRI reveals linear footprint hypointensities containing horizontal striations (arrows) within the pons. Departments of Ophthalmology (JAL, WLB, JSP, MCB) and Neurology (WS, MCB), Mayo Foundation, Mayo Clinic, Rochester, Minnesota. Supported in part by an unrestricted grant from Research to Prevent Blindness, New York, NY and Mayo Foundation, Rochester, MN. M. C. Brodsky is the Knights Templar Professor of Ophthalmology. Address correspondence to Michael C. Brodsky, MD, Mayo Clinic, 200 First Street SW, Rochester, MN 55905; E-mail: brodsky.michael@ mayo.edu Leavitt et al: J Neuro-Ophthalmol 2014; 34: 369-371 369 Photo Essay Section Editor: Timothy J. McCulley, MD Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. ataxia should include a dedicated search for retinal nerve fiber layer thickening, which establishes the diagnosis of ARSACS. Journal of Neuro-Ophthalmology 2014;34:369-371 doi: 10.1097/WNO.0000000000000174 © 2014 by North American Neuro-Ophthalmology Society A39-year-old man of Italian and Irish descent was referred for a long-standing history of clumsiness and gait abnor-malities. At 3 years, his left foot began to turn inward. He was noted to have clumsiness in his hands and legs at school. His handwriting was always poor. This clumsiness progressed until it became difficult for him to run in high school. He developed stiffness and cramps in both legs in his 20s, and his calves and thighs became hypertrophied secondary to spastic-ity. His gait continued to deteriorate, necessitating the use of a cane by age 30. His legs gradually began to atrophy. In his early 20s, he noted numbness of his hands and feet. At age 37, he developed symptoms of urinary urgency, urine leakage, and incomplete bladder emptying. He believed that his speech had always been fine, and he denied vision problems. There was no family history of similar symptoms. On neurological examination, there was ataxic dysarthria with a mild spastic component. Motor examination of the upper extremities showed hypertrophy of deltoid and biceps muscles as well as weakness and atrophy of intrinsic hand muscles. In the lower extremities, there was moderate weakness and atrophy of proximal muscles and severe weakness and atrophy of muscles below the knees. There were occasional fasciculations in thigh and intrinsic hand muscles. Reflexes were hypoactive to absent. There was a mild spastic catch in both lower extremities. He had pes cavus and hammer toes. Sensory testing revealed significant length-dependent sensory impairment to large and small fiber modalities. The patient was unable to walk unassisted; his gait was characterized by ataxia, proximal spasticity, and bilateral foot drop. Coordination could not be adequately assessed in the lower extremities because of the degree of weakness, but there was dysmetria, intention tremor, and irregular rapid alternating movements in the upper extremities. Visual acuity was 20/20 in each eye. Both pupils were briskly reactive with no relative afferent pupillary defect. Extraocular movements were full, with gaze-evoked nystag-mus, saccadic pursuit movements and square wave jerks. Retinal examination disclosed prominent striations of the peripapillary retinal nerve fiber layer of both eyes (Fig. 1A). Optical coherence tomography (OCT) confirmed thicken-ing of the peripapillary nerve fiber layer (Fig. 1B). Magnetic resonance imaging (MRI) showed atrophy of the cerebellar vermis (Fig. 1C) and linear "footprint" hypointensities with pontine striations (Fig. 1D). Genetic testing for autosomal dominant spinocerebellar ataxias was negative. Testing for Friedreich ataxia and selected hereditary spastic paraplegias (SPG3A and 4) was also negative. Sequence analysis of the spastic ataxia of Charlevoix-Saguenay gene (SACS) on chromosome 13 (at Klinisch-Genetisch Centrum Nijmegen) revealed 2 different mutations: A mis-sense mutation c3932T . A (p.Met1311Lys) was found, which is a nonconservative change previously described in an affected consanguineous family with the homozygous state. In addition, an in-frame deletion in exon 9: c.10651 10656del (p.Met3551 Leu3552del) was found, which is a deletion of 2 highly conserved amino acids. His mother was also tested and has the deletion mutation; his father is assumed to have carried the other mutation. In 1978, Bouchard et al (1) first described the clinical signs of autosomal recessive spastic ataxia of Charlevoix- Sagueney (ARSASC). This neurodegenerative disorder is characterized by progressive clumsiness, gait dysfunction, spasticity, dysarthria, muscle weakness, and peripheral amyotrophy (1,2). The combination of spasticity during early development and distal denervation in the lower limbs, which develops later in life lead to foot deformities including clawing, equinus, equinovarus, and club feet (1,2). Bouchard et al (1) noted that all patients had a ".striking and markedly increased visibility of the retinal nerve fibers, mainly in the papillomacular area" bearing some resemblance to that seen in Leber hereditary optic neuropathy. Although isolated thickening and segmentation of the retinal nerve fiber layer was documented in most studies (2-5), focal peripapillary myelination was docu-mented in one patient (4). Saccadic horizontal smooth pur-suit movements and supranuclear vertical gaze deficits have also been described (5). Although affected patients are often visually asymptom-atic, one study documented a delay in the P100 wave of the visual evoked response in all patients (5). OCT has con-firmed thickening of the peripapillary nerve fiber layer, which correlates with the peripapillary retinal nerve fiber layer striations seen clinically (3,4,6-8). OCT does not show the shadowing produced by retinal nerve fiber layer myelination (4). Although this retinal segmentation has been attributed to ganglion cell and nerve fiber layer hyper-plasia (7), a more physiologic mechanism has recently been proposed (8,9). The SACS gene that encodes sacsin, one of the largest proteins in the human genome, is been localized to chromosome 13q12.12 (2,7,10). Depletion of the sacsin protein has been shown to disrupt mitochondrial axonal transport in a transgenic knockout mouse model (8) leading Yu-Wai-Man et al (9) to propose that axonal stasis within the long axons of retinal ganglion cells could produce the diffuse peripapillary retinal nerve fiber layer thickening. Sev-eral magnetic resonance studies have documented atrophy of the superior cerebellar vermis in combination with a char-acteristic footprint linear hypointensities in the pons, which are segmented by numerous irregular fine striations on T2- and T2-fluid-attenuated inversion recovery images (11-13). Lipofuscin deposits are found in cerebellar cortex and in 370 Leavitt et al: J Neuro-Ophthalmol 2014; 34: 369-371 Photo Essay Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. skin (5). Advanced stages of the disease can show progressive atrophy of the cerebellar hemispheres and spinal cord (11). ARSACS was once believed to be rare outside of Quebec, Canada. However, it is now considered to be one of the most common forms of cerebellar ataxia (14,15). Children initially present with ataxia, alone or in combination with pyramidal tract dysfunction (often hyperreflexia rather than spasticity). Because children may not demonstrate clinical signs of neuropathy (15), neuro-ophthalmologic examina-tion of children with cerebellar ataxia should include a ded-icated search for retinal striations, which are seen in children younger than 3 years (A. Duquette, MD, written commu-nication). Our patient epitomizes the adult presentation, wherein the findings of retinal and pontine striations can establish the clinical diagnosis. REFERENCES 1. Bouchard JP, Barbeau A, Bouchard R, Bouchard RW. Autosomal recessive spastic ataxia of Charlevoix-Saguenay. Can J Neurol Sci. 1978;5:61-69. 2. Bouchard JP, Richter A, Mathieu J, Brunet D, Hudson TJ, Morgan K, Melancon SB. Autosomal recessive spastic ataxia of Charlevoix-Saguenay. Neuromuscul Disord. 1998;8:474-479. 3. Desserre J, Devos D, Sautiere BG, Debruyne P, Santorelli FM, Vuillaume T, Defoort-Dhellemmes S. Thickening of peripapillary retinal fibers for the diagnosis of autosomal recessive spastic ataxia of Charlevoix-Saguenay. Cerebellum. 2011;10:758-762. 4. Vingolo EM, Di Fabio R, Salvatore S, Grieco G, Bertini E, Leuzzi V, Nesti C, Filla A, Tessa A, Pirelli F, Santorelli FM, Casali C. Myelinated retina fibers in autosomal recessive spastic ataxia of Charlevoix-Saguenay. Eur J Neurol. 2011;18:1187-1190. 5. Stevens JC, Murphy SM, Davagnanam I, Phadke R, Anderson G, Nethisinghe S, Bremner F, Giunti P, Reilly MM. The ARSACS phenotype can include supranuclear gaze palsy and skin lipofuscin deposits. J Neurol Neurosurg Psychiatry. 2013;84:114-115. 6. Gazulla J, Vela AC, Marin AM, Pablo L, Santorelli FM, Benavente I, Modrego P, Tintore M, Berciano J. Is the ataxia of Charlevoix-Saguenay a developmental disease? Med Hypotheses. 2011;77:347-352. 7. Garcia-Martin E, Pablo LE, Gazulla J, Vela A, Larrosa JM, Polo V, Marques ML, Alfaro J. Retinal segmentation as noninvasive technique to demonstrate hyperplasia in ataxia of Charlevoix-Saguenay. Invest Ophthalmol Vis Sci. 2013;54:7137-7142. 8. Girard M, Lariviere R, Parfitt DA, et al. Mitochondrial dysfunction and Purkinje cell loss in autosomal recessive spastic ataxia of Charlevoix-Saguenay (ARSACS). Proc Natal Acad Sci U S A. 2012;109:1661-1666. 9. Yu-Wai-Man P, Pyle A, Griffin H, Santibanez-Korev M, Horvath R, Chinnery PF. Abnormal retinal thickening is a common feature among patients with ARSACS-related phenotypes. Br J Ophthalmol. 2014;98:711-713. 10. Ogawa T, Takiyama Y, Sakoe K, Mori K, Namekawa M, Shimazaki H, Nakano I, Nishizawa M. Identification of a SACS gene missense mutation in ARSACS. Neurology. 2004;62:107-109. 11. Martin MH, Bouchard JP, Sylvain M, St-Onge O, Truchon S. Autosomal recessive spastic ataxia of Charlevoix-Saguenay: a report of MR imaging in 5 patients. AJNR Am J Neuroradiol. 2007;28:1606-1608. 12. Synofzik M, Soehn AS, Gburek-Augustat J, Schicks J, Karle KN, Schule R, Haack TB, Schoning M, Biskup P, Rodnik- Schoneborn S, Senderek J, Hoffmann KT, MacLeod P, Schwarz J, Bender B, Kruger S, Kreuz F, Bauer P, Schols L. Autosomal recessive spastic ataxia of Charlevoix Saguenay (ARSACS): expanding the genetic clinical and imaging spectrum. Orphanet J Rare Dis. 2013;8:41. 13. Mignarri A, Tessa A, Carluccio MA, Rufa A, Storti E, Bonelli G, Marcotulli C, Santorelli FM, Leonardi L. Cerebellum and neuropsychiatric disorders: insights from ARSACS. Neurol Sci. 2014;35:95-97. 14. Vermeer S, Meijer RP, Pijl BJ, Timmermans J, Cruysberg JR, Bos MM, Schelhaus HJ, van de Warrenburg BP, Knoers NV, Scheffer H, Kremer B. ARSACS in the Dutch population: a frequent cause of early-onset cerebellar ataxia. Neurogenetics. 2008;9:207-214. 15. Duquette A, Brais B, Bouchard JP, Mathieu J. Clinical presentation and early evolution of spastic ataxia of Charlevoix- Saguenay. Mov Disord. 2013;14:2011-2014. Leavitt et al: J Neuro-Ophthalmol 2014; 34: 369-371 371 Photo Essay Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. |