| OCR Text |
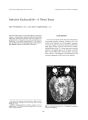
Show Journal of Neuro- Ophthalmotogy 16( 4): 277- 280, 1996. 1996 Lippincott- Raven Publishers, Philadelphia Superficial Siderosis and Episodic Fourth Nerve Paresis Report of a Case with Clinical and Magnetic Resonance Imaging Findings Masato Hashimoto, M. D., and William F. Hoyt, M. D. We describe a patient with superficial siderosis who had an episodic unilateral fourth nerve paresis. The superficial siderosis was caused by small repeated intraventricular hemorrhages from a periventricular cavernous angioma. T2- weighted magnetic resonance images demonstrated a rim of low signal intensity at the brain surface, characteristic of hemosiderin deposition. These low-signal- intensity deposits included the dorsal brain stem around the anterior medullary velum. We suggest that the hemosiderin deposits affected the proximal portion of the fourth nerve where it contains central myelin and that this in some way caused unstable conduction of nerve impulses through the nerve. Key Words: Superficial siderosis- Hemosiderin- Intermittent diplopia- Magnetic resonance imaging. Superficial siderosis of the CNS is a chronic condition in which hemosiderin is deposited on the surface layer of the brain, cranial nerves, and spinal cord. After the first neuropathologic description of neuronal and glial changes in superficial siderosis by Noetzel ( 1) in 1940, it was gradually recognized that superficial siderosis could produce chronic neurologic illness consisting of progressive myelopathy, ataxia, and hearing loss. Once magnetic resonance ( MR) imaging became widely available, clinical reports of superficial siderosis increased sharply ( 2- 9). Because of the paramagnetic effect of hemosiderin in T2- weighted images, superficial siderosis produces a strikingly distinctive black rim ( of low signal intensity) around the brain and brain stem ( 10). To this date no author has suggested that neuro-ophthalmological signs or symptoms should be included among the clinical manifestations of superficial siderosis. We describe episodic diplopia from intermittent trochlear nerve dysfunction in a patient with superficial siderosis confirmed by MR imaging. Manuscript received June 20, 1995; accepted September 1, 1995. From the Neuro- Ophthalmology Unit, Departments of Ophthalmology, Neurological Surgery, and Neurology, School of Medicine, University of California, San Francisco, California, U. S. A. Address correspondence and reprint requests to Dr. William F. Hoyt, University of California, San Francisco, 533 Parnassus Avenue, Room U- 521, San Francisco, CA 94143- 0350, U. S. A. CASE REPORT A 42- year- old woman presented with episodic vertical diplopia. More than 9 years earlier, she had had right- side tinnitus accompanied by headache and leg pain that lasted about 12 days. The pain was precipitated by postural changes from lying to sitting and from sitting to standing. Three years ago, on arising one morning, she had noted that her vision was tilted 45°. When she tried to regain her equilibrium, she lost consciousness for about 30 s. Subsequently, she experienced episodes of severe vertigo and nausea, which re- 277 278 M. HASHIMOTO AND W. F. HOYT solved in 3- 4 days. In February 1994, the patient noted the onset of vertical diplopia while hiking. Within 10 min, single binocular vision returned. Since then, episodes of vertical diplopia have gradually increased in frequency. Neuro- ophthalmologic examination showed visual acuity of 20/ 20 in each eye. The patient had torsional diplopia when she looked downward to the right and when her head was tilted to the left. These findings were consistent with a variable left fourth nerve paresis. T2- weighted MR images showed a rim of low signal intensity around the medulla, pons, and midbrain, particularly at the level of the colliculi, presumably caused by hemosiderin deposits ( Figs. 1 and 2). The T2 images also showed low signal intensity around the intraconal segments of both optic nerves ( Fig. 1). These abnormalities were not apparent on computerized tomography scans or Tl- weighted MR images. Tl- and T2- weighted MR images also showed a small, round, well-demarcated left- side periventricular mass with mixed signal intensity ( Fig. 3), consistent with a cavernous angioma or a cryptic vascular malformation. DISCUSSION Histologically, superficial siderosis consists of intra- and extracellular hemosiderin deposits in the FIG. 1. T2- weighted MR image ( 1.5 T; 2,000/ 90 ms) shows a rim of low signal intensity surrounding the midbrain at the level of superior colliculus. Note especially the dark thick rim of the dorsolateral midbrain surface and the low signal intensity around the intraconal segments of both optic nerves. FIG. 2. T2- weighted MR image shows a rim of low signal intensity compatible with hemosiderin deposits around the upper pons. subpial tissue of the brain and spinal cord and in the ependymal and subependymal lining of the ventricular system. The hemosiderin deposits are thought to be caused by episodic subarachnoid or intraventricular bleeding from capillary or venous sources located anywhere along the cerebrospinal axis. In a review of 40 cases of superficial siderosis FIG. 3. T2- weighted MR image shows a small, round, well- demarcated, left- side periventricular mass with a mixed- signal- intensity mass, consistent with a cavernous angioma or a cryptic vascular malformation. / Neuro- Ophthalmol, Vol. 16, No. 4, 1996 SUPERFICIAL SIDEROSIS AND TROCHLEAR PALSY 279 of the CNS ( 11), the presumed bleeding source was determined in only 26 cases. The most common causes were brain tumors, especially ependymomas ( 12- 14), followed by hemispherectomy ( 15), arteriovenous malformations ( 14), and subdural hematomas ( 14,15). In our patient, the cause of superficial siderosis was thought to be small, repeated intraventricular hemorrhages from a periventricular cavernous angioma. After subarachnoid or intraventricular bleeding, most red blood cells in the cerebrospinal fluid undergo lysis and phagocytosis by macrophages originating from reticuloendothelial cells lining the subarachnoid space. Hemoglobin is converted to hemosiderin and then removed from the subarachnoid space. When chronic bleeding occurs, phagocytosis of the constituents of red blood cells may be incomplete, resulting in the deposition of hemosiderin in the CNS. In a histochemical study using a rabbit model of superficial siderosis, Koeppen et al. ( 14,16) demonstrated that hemosiderin deposits on the surface of the CNS are strongly related to the phagocytosis of microglia of the CNS. Superficial siderosis selectively involves the surfaces of the cerebellum, brain stem, spinal cord, and the first, second, and eighth cranial nerves. Koeppen et al. ( 16) proposed that the vulnerability of the cerebellar molecular layer probably relates to the abundance of ferritin- reactive microglia and the presence of Bergmann glia, including ferritin-repressor protein, which accelerates ferritin biosynthesis. The selective vulnerability of the first and second nerves is explained by the fact that their fibers are invested by central myelin and glia. However, there have been only a few reports of superficial siderosis causing olfactory or visual impairments ( 17- 19). Our patient had normal visual function even though T2- weighted MR images demonstrated superficial low signal intensity on both optic nerves. The eighth nerve is believed to be especially susceptible to the effects of hemosiderin because its transitional zone from central to peripheral myelin extends almost to the internal acoustic meatus ( 20). The transient tilted vision and tinnitus in our patient might have been caused by hemosiderin effects on the subarachnoid segment of the eighth nerve; however, T2- weighted MR imaging showed no obvious deposits in this area. Anatomically, the fourth nerve nucleus is located at the pontomesencephalic junction at the level of the inferior colliculus, just ventrolateral to the cerebral aqueduct. Its fibers course posteroin-feriorly around the aqueduct and decussate within the anterior medullary velum just below the inferior colliculus before they run ventrolaterally around the brain stem in the ambient cistern. Fra-her et al. ( 21) observed histopathologically the transitional zone between central and peripheral myelin in the fourth nerve and noted a distally tapering projection of central myelin extending into the subarachnoid segment of the nerve for a mean distance of 296 ± 76 | xm ( Fig. 4). Hemosiderin deposits in the fourth nerve would occur at the more central portion of this glial transitional zone. In our patient, MR images demonstrated prominent low signal intensity of the dorsal brain stem surface, including the anterior medullary velum, suggesting dense hemosiderin deposits along the proximal portion of the fourth nerve. We have no explanation for the unilaterality of the episodic variations in our patient's fourth nerve function. FIG. 4. The transitional zone between central and peripheral myelin in the fourth nerve; T, transitional zone; IV, the fourth nerve; M, dorsal midbrain; D, dorsal; V, ventral; striped, central tissue; shaded, peripheral tissue. D T V / Neuro- Ophthalmol, Vol. 16, No. 4, 1996 280 M. HASHIMOTO AND W. F. HOYT REFERENCES 1. Noetzel H. Diffusion von Blutfarbstoff in der inneren Rand-zone und ausseren Oberfluache des Zentralnervensystems bei subarachnoidaler Blutung. Arch Psychiatr Nervenkr 1940; 111: 129- 38. 2. Janss AJ, Galetta SL, Freese A, et al. Superficial siderosis of the central nervous system: magnetic resonance imaging and pathological correlation. Case report. / Neurosurg 1993; 79: 756- 60. 3. Willeit J, Aichner F, Felber S, et al. Superficial siderosis of the central nervous system: report of three cases and review of the literature. / Neurol Sci 1992; 111: 20- 4. 4. Pyhtinen J, Paakko E, Ilkko E. Superficial siderosis in the central nervous system. Neuroradiology 1995; 37: 127- 8. 5. Grunshaw ND, Blanshard KS, Hussain SS, Grace ARH. Superficial siderosis of the central nervous system- diagnosis by magnetic resonance imaging. Clin Radiol 1993; 48: 186- 8. 6. Steven MP, Susan AW. Superficial siderosis of the central nervous system. A neglected cause of sensorineural hearing loss. Otolaryngol Head Neck Surg 1992; 107: 69- 77. 7. Daniel D, Bracchi M, Riva A, Duca S, Bradac GB. Superficial siderosis of the central nervous system: neuroradiolog-ical evaluation of two cases. Eur Neurol 1992; 32: 270- 3. 8. Kwartler JA, Cruz ADL, Lo WWM. Superficial siderosis of the central nervous system. Ann Otol Rhinol Laryngol 1991; 100: 249- 50. 9. Bourgouin PM, Tampieri D, Melancon D, Carpio R, Ethier R. Superficial siderosis of the brain following unexplained subarachnoid hemorrhage: MRI diagnosis and clinical significance. Neuroradiology 1992; 34: 407- 10. 10. Bracchi M, Savoiardo M, Triulzi F, et al. Superficial siderosis of the CNS: MR diagnosis and clinical findings. AJNR 1993; 14: 227- 36. 11. Steven I, Petersen D, Grodd W, Poremba M, Dichgans J. Superficial siderosis of the central nervous system. A 37- year follow- up of a case and review of the literature. Eur Arch Clin Neurosci 1991; 241: 57- 60. 12. Gomori JM, Grossman RI, Bilanuik LT, Zimmerman RA, Goldberg HI. High- field MR imaging of superficial siderosis of the central nervous system. / Comput Assist Tomogr 1985; 9: 972- 5. 13. Tomlinson BE, Walton JN. Superficial haemosiderosis of the central nervous system. / Neurol Neurosurg Psychiatry 1964; 27: 332- 9. 14. Koeppen AH, Dentinger MP. Brain hemosiderin and superficial siderosis of the central nervous system. / Neuro-pathol Exp Neurol 1988; 47: 249- 70. 15. Hughes JT, Oppenheimer DR. Superficial siderosis of the central nervous system. A case report on nine cases with autopsy. Acta Neuropathol ( Bed) 1969; 13: 56- 74. 16. Koeppen AH, Dickson AC, Chu RC, Thach RE. The pathogenesis of superficial siderosis of the central nervous system. Ann Neurol 1993; 34: 646- 53. 17. Sherwin I, Toll K. Superficial hemosiderosis of the central nervous system. Dis Nerv Syst 1972; 6: 413- 7. 18. Lewy FH, Govons SR. Hemochromatotic pigmentation of the central nervous system. / Neuropathol Exp Neurol 1942; 1: 129- 38. 19. Garcin R, Lapresle J. Sur un cas de surcharge ferrique du systeme nouveaux central. Rev Neurol 1957; 97: 417- 32. 20. Tarlov IM. Structure of the nerve root I. Nature of the junction between the central and peripheral nervous system. Arch Neurol Psychiatry 1937; 37: 555- 83. 21. Fraher JP, Smiddy PF, O'Sullivan VR. The central-peripheral transitional regions of cranial nerves. Trochlear and abducent nerves. / Anat 1988; 161: 115- 23. / Neuro- Ophthalmol, Vol. 16, No. 4, 1996 |