| OCR Text |
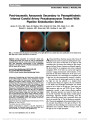
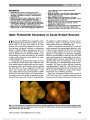
Show intractable headaches and had worsening papilledema. Open-ing pressure on repeat lumbar puncture was 44 cm H2O. At this point, the patient's mother reported that the child had OSA, which was diagnosed 2 years previously. Polysomnographic studies had demonstrated severe OSA with a respiratory disturbance index of 21 events per hour. The patient was irregularly using a continuous positive air-way pressure machine. On physical examination, the patient was found to have hypertrophic tonsils and adenoids with complete obstruc-tion of her nasopharynx. She underwent adenotonsillec-tomy and postoperatively reported immediate relief of her headaches. At her last visit, she had complete resolution of all symptoms referable to IIH and subjective improvement in her sleeping pattern. Recent studies have suggested a potential relationship between IIH and OSA (2-6). The nocturnal hypoxemia and hypercarbia present during apneic episodes cause ce-rebral vasodilation and secondary increased ICP (7). OSA may be a risk factor in the worsening of IIH, the impor-tance of which is compounded by the fact that the 2 con-ditions frequently coexist in patients with obesity. Screening for papilledema is recommended in all patients with OSA who have visual symptoms (8,9). In children who are found to have OSA and IIH, airway obstruction caused by adenotonsillar enlargement should be consid-ered. We are not aware of previously reported cases where the removal of adenotonsillar tissue led to significant im-provement in OSA and resolution of intracranial hypertension. Evelyne Kalyoussef, MD Nneka O. Brooks, MD Huma Quraishi, MD Roger Turbin, MD Larry Frohman, MD Departments of Otolaryngology-Head and Neck Surgery (EK, HQ), Ophthalmology and Visual Sciences (NO, RT, LF), and Neurology and Neurosciences (LF), Rutgers New Jersey Medical School, Newark, New Jersey The authors report no conflicts of interest. Presented at Triological Society Combined Sections Meeting, January 27-29, 2011, Scottsdale, AZ. REFERENCES 1. Wall M. Idiopathic intracranial hypertension and the idiopathic intracranial Hypertension Treatment Trial. J Neuroophthalmol. 2013;33:1-3. 2. Quinn AG, Gouws P, Headland S, Oades P, Pople I, Taylor D, Benton JS, Buncic JR, Henderson J, Fleming P. Obstructive sleep apnea syndrome with bilateral papilledema and vision loss in a 3-year-old child. J AAPOS. 2008;12:197-199. 3. Hanigan WC, Zallek SN. Headaches, shunts, and obstructive sleep apnea: report of two cases. Neurosurgery. 2004;54:764- 768; discussion 768-760. 4. Marcus DM, Lynn J, Miller JJ, Chaudhary O, Thomas D, Chaudhary B. Sleep disorders: a risk factor for pseydotumor cerebri. J Neuroophthalmol. 2001;21:121-123. 5. Lee A. Pseudotumor and sleep apnea. J Neuroophthalmol. 2001;21:235. 6. Wall M, Purvin V. Idiopathic intracranial hypertension in men and the relationship to sleep apnea. Neurology. 2009;72:300-301. 7. Purvin VA, Kawasaki A, Yee RD. Papilledema and obstructive sleep apnea syndrome. Arch Ophthalmol. 2000;118:1626-1630. 8. Waller EA, Bendel RE, Kaplan J. Sleep disorders and the eye. Mayo Clin Proc. 2008;83:1251-1261. 9. Jindal M, Hiam L, Raman A, Rejali D. Idiopathic intracranial hypertension in otolaryngology. Eur Arch Otorhinolaryngol. 2009;266:803-806. A Limited Form of Neuromyelitis Optica With a Lesion of the Fourth Nerve Nucleus We read with great interest the review article "Neuro-myelitis optica" by Morrow and Wingerchuk (1). The authors referred to several reports of abnormal eye move-ments in association with brainstem lesions in NMO (2-4). We evaluated a patient with a limited form of NMO with a fourth nerve palsy. A 62-year-old woman with a history of numbness in the legs, nausea, and repeated vomiting presented with vertical diplopia and numbness in the left arm and chest. Neuro-ophthalmic testing demonstrated visual acuity of 20/20 in both eyes, full visual fields, and normal ophthalmoscopy. The patient had a right hypertropia that increased in left gaze and head tilt to the right. Ocular motility was otherwise unremarkable. Magnetic resonance imaging (MRI) showed hyperin-tense areas in the midbrain, pons, and medulla (Fig. 1) and in the spinal cord from C5 to T1 (Fig. 2). NMO antibody was positive, whereas acetylcholine receptor, nuclear, SS-A, and SS-B antibodies were all negative. Cerebrospinal fluid analysis revealed no oligoclonal bands. The patient was diagnosed with a limited form of NMO due to the presence of NMO antibody and longitudinally extensive spinal cord lesions. She was treated with intravenous methylprednisolone pulse ther-apy (1,000 mg daily for 3 days) followed by oral prednisolone (10 mg/day with gradual taper). Two months after the third pulse, vertical diplopia had almost 414 Letters to the Editor: J Neuro-Ophthalmol 2013; 33: 412-423 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. totally resolved with a decrease of the hyperintense areas on MRI. Our patient with a right fourth nerve palsy had a form of NMO, a subtype of NMO spectrum disorder. Reports of cases of NMO or NMO spectrum disorder with ophthal-moplegia are rare. Gilmore et al (5) described a patient with Parinaud syndrome, in which MRI abnormalities were found in periaqueductal gray matter. Shinoda et al (3) found a mid-brain tegmentum lesion adjacent to the aqueduct on MRI causing wall-eyed bilateral internuclear ophthalmoplegia syn-drome in a patient with NMO spectrum disorder. In our patient, MRI showed a high-intensity lesion in the area of the fourth nerve nucleus causing vertical diplopia. In addition, our patient had a history of nausea and vomiting likely due to NMO involving the medullary floor of the fourth ventricle and area postrema (6,7). Mikihide Ogasawara, MD Tsutomu Sakai, MD, PhD Department of Ophthalmology, The Jikei University School of Medicine, Nishishinbashi, Minato-ku, Tokyo, Japan Yu Kono, MD, PhD Department of Neurology, The Jikei University School of Medicine, Nishishinbashi, Minato-ku, Tokyo, Japan Keigo Shikishima, MD, PhD Hiroshi Tsuneoka, MD, PhD Department of Ophthalmology, The Jikei University School of Medicine, Nishishinbashi, Minato-ku, Tokyo, Japan The authors report no conflicts of interest. REFERENCES 1. Morrow MJ, Wingerchuk D. Neuromyelitis optica. J Neuroophthalmol. 2012;32:154-166. 2. Hage R Jr, Merle H, Jeannin S, Cabre P. Ocular oscillations in the neuromyelitis optica spectrum. J Neuroophthalmol. 2011;31:255-259. 3. Shinoda K, Matsushita T, Furuta K, Isobe N, Yonekawa T, Ohyagi Y, Kira J. Wall-eyed bilateral internuclear ophthalmoplegia (WEBINO) syndrome in a patient with neuromyelitis optica spectrum FIG. 1. A. Sagittal T2 magnetic resonance imaging (MRI) shows hyperintense lesions in the midbrain and pons (arrowheads). Axial FLAIR MRI reveals hyperintense lesions in the midbrain (B, arrowhead), pons (C, arrowhead), and left side of the medulla oblongata (D, arrowhead). FIG. 2. Sagittal T2 spine magnetic resonance imaging demonstrates a hyperintense lesion extending for five ver-tebral segments (C5 to T1; arrowheads). Letters to the Editor: J Neuro-Ophthalmol 2013; 33: 412-423 415 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. disorder and anti-aquaporin-4 antibody. Mult Scler. 2011;17:885-887. 4. Wang KC, Lee CL, Chen SY, Lin KH, Tsai CP. Prominent brainstem symptoms/signs in patients with neuromyelitis optica in a Taiwanese population. J Clin Neurosci. 2011;18:1197-1200. 5. Gilmore CP, Jacob A, Evangelou N. A case of neuromyelitis optica with gadolinium-enhancing brain lesions and Parinaud syndrome. Arch Neurol. 2009;66:140- 141. 6. Patel V, Griffith NC, Blackwood E, Dias M, Cordato DJ. Spectrum disorder of neuromyelitis optica in a patient presenting with intractable vomiting and hiccups, transverse myelitis and acute encephalopathy. J Clin Neurosci. 2012;19:1576-1578. 7. Popescu BF, Lennon VA, Parisi JE, Howe CL, Weigand SD, Cabrera-Gómez JA, Newell K, Mandler RN, Pittock SJ, Weinshenker BG, Lucchinetti CF. Neuromyelitis optica unique area postrema lesions: nausea, vomiting, and pathogenic implications. Neurology. 2011;76:1229-1237. Spontaneous Cerebrospinal Fluid Otorrhea and Rhinorrhea in Idiopathic Intracranial Hypertension Patients The article by Rosenfeld et al (1) described cerebro-spinal fluid (CSF) leaks caused by chronically in-creased intracranial pressure (ICP) in 4 patients with idiopathic intracranial hypertension (IIH). The impres-sion given by the authors is that spontaneous CSF leaks occur with equal frequency in the settings of normal and increased ICP. However, evidence indicates that the ma-jority of spontaneous CSF leaks are associated with in-tracranial hypertension. Spontaneous CSF leaks traditionally have had a high recurrence rate after surgical repair (25%-87%), compared to less than 10% for most other etiologies (2-4). Identification of this underlying etiology has led to the widespread use of acetazolamide and, in some cases, permanent CSF diversion to control the ICP. In a prospective evaluation of more than 5 years, 46 patients (average age, 51 years) with a cumulative 56 spontaneous CSF leaks were treated by the senior author (5). The data presented in the study provided concrete evidence that the majority of spontaneous CSF leaks are secondary to intracranial hypertension. Lumbar drain pressure measurements averaged 32.3 6 9.0 cm H2O and demographics mirrored IIH, where a large proportion of the patient cohort consisted of obese middle-aged women. Successful treatment of ele-vated ICP in combination with endoscopic repair can provide high success rates (93% primary and 100% secondary). Michael S. Vaphiades, DO Neel K. Ranganath, BS Bradford A. Woodworth, MD Department of Ophthalmology, Neurology, and Neurosurgery (MSV), University of Alabama at Birmingham, Birmingham, Alabama; and Department of Surgery, Division of Otolaryngology, Head & Neck Surgery (NKR, BAW), University of Alabama at Birmingham, Birmingham, Alabama B. A. Woodworth is a consultant for Gyrus, ArthroCare ENT, and Cook Medical. The other authors report no conflicts of interest. ACKNOWLEDGMENTS This work was supported in part by an unrestricted grant from the Research to Prevent Blindness, Inc, New York, NY. REFERENCES 1. Rosenfeld E, Dotan G, Kimchi TJ, Kesler A. Spontaneous cerebrospinal fluid otorrhea and rhinorrhea in idiopathic intracranial hypertension patients. J Neuroophthalmol. 2013;33:113-116. 2. Woodworth BA, Palmer JN. Spontaneous cerebrospinal fluid leaks. Curr Opin Otolaryngol Head Neck Surg. 2009;17:59-65. 3. Woodworth B, Prince A, Chiu AG, Cohen NA, Schlosser RJ, Bloger WE, Kennedy DW, Palmer JN. Spontaneous CSF leaks: a paradigm for definitive repair and management of intracranial hypertension. Otolaryngol Head Neck Surg. 2008;138:715- 720. 4. Schlosser RJ, Woodworth BA, Wilensky EM, Grady MS, Bolger WE. Spontaneous cerebrospinal fluid leaks: a variant of benign intracranial hypertension. Ann Otol Rhinol Laryngol. 2006;115:495-500. 5. Chaaban MR, Illing E, Riley KO, Woodworth BA. Spontaneous cerebrospinal fluid leak repair: a five-year prospective evaluation. Laryngoscope. [published ahead of print June 20, 2013] doi: 10.1002/lary.24160. 416 Letters to the Editor: J Neuro-Ophthalmol 2013; 33: 412-423 Letters to the Editor Copyright © North American Neuro-Ophthalmology Society. Unauthorized reproduction of this article is prohibited. |