| OCR Text |
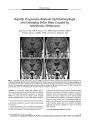
Show ORIGINIAL CONTRIBUTION Pitfalls in the Diagnosis of Mitochondrial Encephalopathy with Lactic Acidosis and Stroke- Like Episodes Iris Ben- Bassat Mizrachi, MD, Diana Gomez- Hassan, MD, PhD, Mila Blaivas, MD, PhD, and Jonathan D. Trobe, MD Abstract: We describe a patient with genetically- and biochemically- proven mitochondrial encephalopathy with lactic acidosis and stroke- like episodes ( MELAS) who was initially misdiagnosed as having had multiple ischemic strokes in part because the clinical presentation appeared to be acute, the MRI of lesions showed restricted diffusion, and the brain biopsy showed features suggestive of stroke. This report emphasizes the pitfalls in the diagnosis of MELAS and points out the similarities and differences between MELAS and ischemic stroke. (/ Neuro- Ophthalmol 2006; 26: 38- 43) itochondrial encephalomyopathy, lactic acidosis, and stroke- like episodes ( MELAS) has clinical, imaging, and brain biopsy features that resemble those of ischemic stroke. We report a patient with genetically- and biochemically-proven MELAS who was initially misdiagnosed as having had ischemic stroke due to an acute neurologic decline and an MRI scan and brain biopsy interpreted as most consistent with ischemic stroke. We call attention to features that promote diagnostic confusion between MELAS and ischemic stroke. CASE REPORT A 34- year- old man was hospitalized in July 2004 because of fatigue and weakness of the left side of the body for several days and increased lethargy and confusion in the past few weeks. He had a history of insulin- dependent diabetes mellitus, bilateral sensorineural hearing loss, and abuse of alcohol, cocaine, and marijuana. Department of Ophthalmology ( IB- BM, JDT), W. K. Kellogg Eye Center, Departments of Radiology ( DG- H), Pathology ( MB), and Neurology ( JDT), University of Michigan, Ann Arbor, Michigan. Address correspondence to Jonathan D. Trobe, MD, Kellogg Eye Center, 1000 Wall Street, Ann Arbor, MI 48105; E- mail: jdtrobe@ umich. edu Supported in part by a fellowship from the American Physician Fellowship for Medicine in Israel. General physical examination demonstrated short stature and normal vital signs. He was alert and fully oriented. Neurologic examination was otherwise normal apart from reduced strength in the left upper and both lower extremities and diminished deep tendon reflexes throughout. MRI demonstrated a large area of high signal on T2- weighted and FLAIR sequences that involved the right posterior frontal, temporal, parietal, and occipital lobes with midline and uncal shift due to mass effect. Diffusion-weighted images ( DWIs) showed a small area of high signal in the occipital region interpreted as T2 " shine-through" rather than restricted diffusion since the apparent diffusion coefficient ( ADC) value was normal. The regions of signal abnormality extended across vascular boundaries ( Fig. 1, A- C). Tl- weighted imaging showed high signal in the basal ganglia bilaterally ( Fig. ID). The imaging findings were interpreted as consistent with subacute infarction albeit atypical as the signal abnormalities transgressed the domain of a single circumflex cerebral artery. The basal ganglia Tl high signal was attributed to mineralization. Electroencephalography ( EEG) demonstrated bilateral generalized background slowing suggestive of diffuse encephalopathy and profound slowing involving the right hemisphere. The tentative clinical diagnosis was subacute ischemic stroke attributed to diabetes and substance abuse. However, because of his young age, the bilaterality of the EEG abnormalities, and the fact that MRI abnormalities extended beyond a single arterial domain, he underwent brain biopsy. Brain biopsy demonstrated multiple small confluent regions of recent parenchymal damage that contained proliferating small vessels, macrophages, perivascular edema with lymphocytic infiltration and hemosiderin deposits, ischemic neurons, and reactive astrocytes ( Fig. 2). These findings were interpreted as consistent with ischemic infarct. But because they were not entirely specific for infarct, he was treated for herpes simplex encephalitis with intravenous acyclovir for ten days. After discharge to a rehabilitation facility, he was temporarily lost to follow- up. Four months later, he was re- admitted for headache, vomiting, visual deterioration, and confusion developing 38 J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. MELAS J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 FIG. 1. A- D. MRIs from the first admission, July 2004, weeks after onset of left- sided weakness, lethargy, and confusion. A. Axial T2 image shows cortical thickening and increased signal in subjacent white matter of the right temporo- parieto-occipital region. B. Axial FLAIR image shows the same findings. C. Diffusion- weighted image ( DWI) shows a small area of high signal in the occipital region interpreted as T2 " shine- through" rather than restricted diffusion ( apparent diffusion coefficient value was normal). D. Axial pre- contrast T1 shows high signal in the caudate and putamen bilaterally. E- H. MRIs from the second admission, November 2004, days after development of acute confusion and visual loss. E. Axial T2 image shows residual high signal in the right posterior cerebral hemisphere with striking new cortical thickening and subjacent white matter high signal in the left temporo- occipital region. F. Axial FLAIR image shows the same findings. C. DWI shows large area of high signal in the same location interpreted as restricted diffusion. H. Axial pre- contrast T1 again shows high signal in the caudate and putamen bilaterally. I- K. MRIs from second admission, December 2004. I. Axial T2 image shows lessening of high signal in previously affected region. J. Axial FLAIR shows the same findings. K. DWI shows regression of restricted diffusion. L. Non- contrast CT from the second admission, November 2004. It shows high attenuation in the basal ganglia bilaterally. There is a large area of low attenuation in the posterior cerebral hemispheres bilaterally reflecting tissue damage there. 39 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 Ben- Bassat Mizrachi et al FIG. 2. Brain biopsy from the right temporal lobe ( H& E stain) shows small vessel proliferation ( black arrow), edema ( black arrow head), perivascular lymphocytic infiltration ( white arrow head), and reactive astrocytes ( white arrow). over several days. He was disoriented to time and place, did not follow simple commands, had slow and echolalic speech, and demonstrated combative behavior. He was too inattentive to cooperate adequately with complex neurological assessment, but he appeared to have visual acuity no better than finger counting OU and he did not blink to threat. He would not cooperate for confrontation visual field testing. Extraocular movements and alignment were normal, pupils were round and reactive without relative afferent pupillary defect, and fundi were normal. He appeared to have normal strength. Deep tendon reflexes were decreased in all extremities. MRI demonstrated a new left parieto- temporo- occipital lesion with confluent high T2- weighted and FLAIR signal and restricted diffusion. There was right parieto- temporo-occipital encephalomalacia without restricted diffusion ( Fig. 1, E- G). Tl- weighted imaging again showed high signal in the basal ganglia bilaterally which was present on a non- contrast CT ( Fig. 1, H- I). The new imaging abnormalities were considered consistent with acute ischemia in the left cerebral hemisphere attributable to small vessel disease. However, the possibility of mitochondrial disease was now raised, principally because of the basal ganglia signal abnormality. A cerebral angiogram was normal, showing no evidence of vasculitis. A work- up for hypercoagulability and an embolic source was negative. More attention was now paid to the bilateral sensorineural hearing loss, short stature, history of diabetes mellitus, and the fact that imaging abnormalities crossed vascular boundaries. In aggregate, these findings led to a suspicion of MELAS. Lactic acid was found to be elevated at 3.9 mmol/ liter ( normal 0.5- 2.2 mmol/ liter) in blood and 4.8 mmol/ liter in the cerebrospinal fluid ( normal < 3 mmol/ liter), which had an otherwise normal formula. Vastus lateralis muscle needle biopsy showed numerous ragged red fibers ( Fig. 3). Mitochondrial DNA analysis of a peripheral blood leukocyte sample yielded an A- to- G point mutation at base pair 3243, confirming the diagnosis of MELAS. A brain MRI performed three weeks after the second hospital admission demonstrated unchanged T2- weighted and FLAIR high signal in the same region; these lesions no longer had restricted diffusion ( Fig. 1, J- K). DISCUSSION We report a case with a classical clinical presentation of MELAS that was initially misdiagnosed as ischemic stroke. The misdiagnosis was based on three features: 1) a clinical course consisting of rapid decline in neurological function, 2) MRI abnormalities considered compatible with ischemic stroke, and 3) brain biopsy considered compatible with ischemic stroke. Clinical Course On the first admission, our patient presented with evolving symptoms over several weeks. Physicians dismissed the protracted history in favor of the striking imaging findings to reach a diagnosis of stroke. On the second admission, four months later, the presentation was much more acute, leading to an even stronger misimpression of stroke. The tempo of clinical manifestations in MELAS can sometimes lead to a misdiagnosis of stroke. Symptoms are usually subacute in onset but may be as acute as is seen in ischemic stroke ( hence the term " stroke- like" in MELAS). FIG. 3. Skeletal muscle biopsy. Engel's modification of Gomori trichrome stain ( X200) shows " ragged red" muscle fibers (*) typical of mitochondrial diseases. 40 © 2006 Lippincott Williams & Wilkins Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. MELAS J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 In 110 patients with MELAS, Hirano et al ( 1,2) reported acute stroke- like episodes in 14 patients ( 13%). Protracted symptoms tend to precede the acute neurologic decline, but they are often overlooked. Among 40 patients with MELAS, Goto et al ( 3) found that muscle weakness, mental retardation, headache, short stature, and vomiting were present in 50% of patients before the acute stroke- like episodes, but that these manifestations were not sufficiently severe to bring the patient to medical attention. MRI Scan The MRI differentiation between MELAS and ischemic stroke has been a source of confusion. Four distinguishing features of MELAS have been described, but none is entirely specific. Cortical Gray Matter- Based Signal Abnormalities. Cortical gray matter signal abnormalities are reported to be based more common in MELAS than in ischemic stroke. For example, Matthews et al ( 4) described MRI lesions in three MELAS patients that were confined to the cortex and adjacent white matter with relative sparing of the deep white matter. The authors suggested that the gray matter, which is metabolically more active, is more vulnerable than the white matter. In 13 of 14 autopsies of MELAS patients, Hirano et al ( 1,2) found that the lesions were located in the cortex and subcortical white matter with relative sparing of the deep white matter. However, there are also several reports describing predominant involvement of the white matter ( 5,6). In four patients with MELAS, Barkovich et al ( 7) found that two had mainly white matter disease with relative sparing of cerebral cortex " a fact that initially obscured the diagnosis." Thus, the presence of predominantly white matter signal abnormalities should not be construed as excluding MELAS. Lack of Restricted Diffusion. A normal ADC, implying lack of restricted diffusion, is said to occur in MELAS but not in acute stroke, which characteristically shows a low ADC. Several reports ( 8- 13) describe cases of MELAS with a normal or elevated ADC. This differentiating point has become such accepted dogma that it appears in review articles and text books ( 14), as evidenced by the following comment: "... increased ADC values in the appropriate clinical setting must raise suspicion of MELAS" ( 15). However, Wang et al ( 16) have reported a low ADC in a patient with MELAS on an MRI done five days after presentation. Follow- up MRI six days later continued to demonstrate a low ADC, but a scan performed 189 days later showed a normal ADC. Our patient did not have restricted diffusion on MRI during the first hospital admission, but did have restricted diffusion on MRI during the second hospital admission. How can we reconcile these differing findings? On the first admission, the imaging study was performed several weeks after the onset of symptoms, too late to show restricted diffusion. On the second admission, the MRI was performed within days of acute neurological decline. Three weeks later, MRI showed disappearance of the restricted diffusion and normalization of the ADC value ( Fig 1., I- K), exactly as would be seen in ischemic stroke. Thus, the literature claiming that the pattern of restricted diffusion is different in acute stroke and MELAS is misleading. We believe our case to be the first to demonstrate that the evolution of restricted diffusion occurs in MELAS just as it does in ischemic stroke, except that in MELAS, the time of onset of neuronal death is difficult to determine. We presume that previously reported cases that failed to show restricted diffusion had their MRIs performed too late to show it. Signal Abnormalities Extending across Vascular Domains. Unlike most ischemic stroke, MELAS signal abnormalities are generally not restricted to traditional vascular territories. But combined territorial and watershed ischemic strokes may certainly extend across traditional territorial domains of vascular supply. Indeed, clinicians interpreted that our patient's MRI findings were consistent with such a combination. High Tl and Low T2 Signal in the Basal Ganglia. Such signal abnormalities, located predominantly in the globus pallidus and caudate nucleus, are detected in 43%- 53% of patients with MELAS ( 2). Histopathologically, calcium deposits are found in these tissues in 65% ( 2,14,17), generally within small blood vessels ( 1,17- 19). Electron microscopic analysis of the calcified small vessels of the globus pallidus in one MELAS autopsy revealed increased calcium, phosphorus, and iron ( 20). As seen in our patient, these signal abnormalities may be explained by shortening of the Tl relaxation time of the hydrogen protons next to the surface of calcium crystals ( 21- 23). Such signal abnormalities do not typically occur in ischemic stroke, which rarely targets these tissues bilaterally. Instead, they reflect intracellular energy failure from anoxia, toxins, electrolyte disturbances, nutritional disorders, and genetic disorders affecting oxidative phosphorylation ( 24). In essence, mineralization occurs as a marker of metabolic cell death ( dystrophic mineralization). Such signal changes on CT and MRI may also occur as an age- related phenomenon. They were found in 0.36%- 0.6% of 5,000 ( 25) and 12,000 ( 26) CT scans but only in the globus pallidus. Thus, such signal changes found in patients under the age of 40 or outside the globus pallidus should not be dismissed. Our patient demonstrated hyperintense Tl signal and low T2 signal on all MRI scans, but on the first MRI scan these characteristics were considered incidental and were upstaged by the overwhelming signal abnormalities in the cerebral white and cortical gray matter. 41 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 Ben- Bassat Mizrachi et al Magnetic resonance spectroscopy ( MRS) may offer an adjunct to MRI in differentiating MELAS from stroke. Reflecting the metabolic consequence of disruption of oxidative phosphorylation, the MRS findings in MELAS include markedly elevated lactate and reduced N- acetylaspartate ( NAA), glutamate, and myo- inositol ( 27,28). MRS can also demonstrate the presence of elevated lactate in normal-appearing brain parenchyma of MELAS patients ( 6,28). The MRS changes in acute schemic stroke are similar to those of MELAS, consisting of elevated lactate and reduced NAA due to lack of perfusion and conversion from oxidative to anaerobic metabolism. In ischemic stroke, however, these MRS changes will be restricted to the ischemic region and would not, as in MELAS, appear in a normal- appearing brain ( 29). Brain Biopsy The third pitfall in our case was the brain biopsy, which was interpreted as consistent with an ischemic infarct. However, the histopathology of mitochondrial encephalomyopathies resembles that of ischemic infarct in showing gross swelling due to edema in the acute stage and cystic changes in the chronic stage. Microscopically, the acute/ subacute stages display neuronal eosinophilia, reactive astrocytosis, infiltration of macrophages and lymphocytes, cytotoxic and vasogenic edema, and proliferation of small blood vessels ( 17). These findings are non- specific results of reduced oxygen, blood flow, or glucose, and malfunction of oxidative phosphorylation. In 10 of 14 MELAS autopsies, Hirano et al ( 2) found changes identical to those seen in ischemic infarcts. However, two features that distinguished MELAS from ischemic stroke were calcification of the basal ganglia, present in 11 of the 14 cases, and ragged red muscle fibers, present in all 12 cases in which muscles were examined. In seven MELAS autopsies, Tanahashi et al ( 30) reported findings similar to those of Hirano et al ( 2). In making the diagnosis of MELAS, the clinician should be aware of the pitfalls that suggest stroke. The clinical course is usually subacute but may be acute, the MRI may show restricted diffusion if performed within the first week of onset of new symptoms, and the brain biopsy may show changes identical to those of stroke. Even so, the diagnosis should be suspected in a patient who is not stroke- prone and has short stature, sensorineural hearing loss, and an MRI that shows altered signal extending across vascular boundaries and Tl high signal in the deep cerebral gray matter. REFERENCES 1. Hirano M, Pavlakis SG. Mitochondrial myopathy, encephalopathy, lactic acidosis and stroke- like episodes MELAS: current concepts. J Child Neurol 1994; 9: 4- 13. 2. Hirano M, Ricci E, Koenigsberger MR. MELAS: an original case and clinical criteria for diagnosis. Neuromasc Disord 1992; 2: 125- 3. 3. Goto Y, Horai S, Matsuoka T, et al. Mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke- like episodes ( MELAS): a correlative study of the clinical features and mitochondrial DNA mutations. Neurology 1992; 42: 545- 50. 4. Matthews PM, Tampieri D, Berkovic ST, et al. Magnetic resonance imaging shows specific abnormalities in MELAS syndrome. Neurology 1991; 41: 1043- 6. 5. Apostolova LG, White M, Moore SA, et al. Deep white matter pathologic features in watershed regions. A novel pattern of central nervous system involvement in MELAS. Arch Neurol 2005; 62: 1154- 6. 6. Castillo M, Kwock L, Green C. MELAS syndrome: imaging and proton MR spectroscopic findings. AJNR Am JNeuroradiol 1995; 16: 233- 9. 7. Barkovich AJ, Good Wy Koch TK, et al. Mitochondrial disorders: analysis of their clinical and imaging characteristics. AJNR Am J Neuroradiol 1993; 14: 1119- 37. 8. Oppenheim C, Galanaud D, Samson Y, et al. Can diffusion weighted magnetic resonance imaging help differentiate stroke from strokelike events in MELAS? J Neurol Neurosurg Psychiatry 2000; 69: 248- 50. 9. Kolb SJ, Costello F, Lee AG, et al. Distinguishing ischemic stroke from stroke- like lesions in MELAS using apparent diffusion coefficient mapping. J Neurol Sciences 2003; 216: 11- 5. 10. Yonemura K, Hasegawa Y, Kimura K, et al. Diffusion- weighted MR imaging in a case of mitochondrial myopathy, encephalopathy, lactic acidosis, and strokelike episodes. AJNR Am J Neuroradiol 2001; 22: 269- 72. 11. Ohshita T, Oka M, Imon Y, et al. Serial diffusion- weighted imaging in MELAS. Neuroradiology 2000; 42: 651- 6. 12. Yoneda M, Maeda M, Kimura H. Vasogenic edema on MELAS: a serial study with diffusion- weighted MR imaging. Neurology 1999; 53: 2182^. 13. Majoie CB, Akkerman EM, Blank C. Mitochondrial encephalomy-opathy: comparison of conventional MR imaging with diffusion-weighted and diffusion tensor imaging: case report. AJNR Am J Neuroradiol 2002; 23: 813- 6. 14. Osborn AG. Metabolic/ degenerative disorders, inherited. In: Osborn AG, Blaser S, Salzman KL. Diagnostic Imaging: Brain. Salt Lake City, Utah: Amirsys Inc.; 2004: 16- 19. 15. Thambisetty M, Newman NJ. Diagnosis and management of MELAS. Expert Rev Mol Diagn 2004; 4: 631^ 4. 16. Wang XY, Noguchi K, Takashima S, et al. Serial diffusion- weighted imaging in a patient with MELAS and presumed cytotoxic edema. Neuroradiology 2003; 45: 640- 3. 17. Powers JM, de Vivo DC. Peroxisomal and mitochondrial disorders. In: Graham DI, Lantos PL, eds. Greenfield's Neuropathology. Vol. I. London: Arnold; 2002: 780- 784. 18. Sparaco M, Bonilla E, DiMauro S, et al. Neuropathology of mitochondrial encephalomyopathies due to mitochondrial DNA defects. J Neuropath Exp Neurol 1993; 52: 1- 10. 19. Prayson RA, Wang N Mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke- like episodes ( MELAS) syndrome: an autopsy report. Arch Pathol Lab Med 1998; 122: 978- 81. 20. Kishi M, Yamamura Y, Kurihara T, et al. An autopsy case of mitochondrial encephalomyopathy: biochemical and electron microscopic studies of the brain. J Neurol Sci 1988; 86: 31^ 0. 21. Anderson JC, Costantino MM, Stratford T. Basal ganglia: anatomy, pathology, and imaging characteristics. Curr Probl Diagn Radiol 2004; 33: 28^ 1. 22. Avrahami E, Cohn DF, Feibel M, et al. MRI demonstration and CT correlation of the brain in patients with idiopathic intracerebral calcification. J Neurol 1994; 241: 381^ k 23. Henkelman RM, Watts JF, Kucharczyk W. High signal intensity in MR images of calcified brain tissue. Radiology 1991; 179: 199- 206. 24. Osborne AG. Diagnostic Neuroradiology. St. Louis, MO: Mosby; 1994: 777. 42 © 2006 Lippincott Williams & Wilkins Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. MELAS J Neuro- Ophthalmol, Vol. 26, No. 1, 2006 25. Brannan TS, Burger AA, Chaudhary MY. Bilateral basal ganglia calcifications visualized on CT scan. J Neurol Neurosurg Psych 1980; 43: 403- 6. 26. Cohen CR, Duchesneau PM, Weinstein MA. Calcification of the basal ganglia as visualized by computed tomography. Radiology 1980; 134: 97- 9. 27. Moller HE, Kurlemann G, Putzler M, et al. Magnetic resonance spectroscopy in patients with MELAS. J Neurol Set 2005; 229- 230: 131- 9. 28. Abe K, Yoshimura H, Tanaka H, et al. Comparison of conventional and diffusion- weighted MRI and proton MR spectroscopy in patients with mitochondrial encephalomyopathy, lactic acidosis, and strokelike events. Neuroradiology 2004; 46: 113- 7. 29. Castillo M, Kwock L, Mukherji SK. Clinical application of proton MR spectroscopy. AJNR Am JNeuroradiol 1996; 17: 1- 15. 30. Tanahashi C, Nakayama A, Yoshida M, et al. MELAS with the mitochondrial DNA 3243 point mutation: a neuropathological study. Acta Neuropathol 2000; 99: 31- 8. 43 Copyright © Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. |